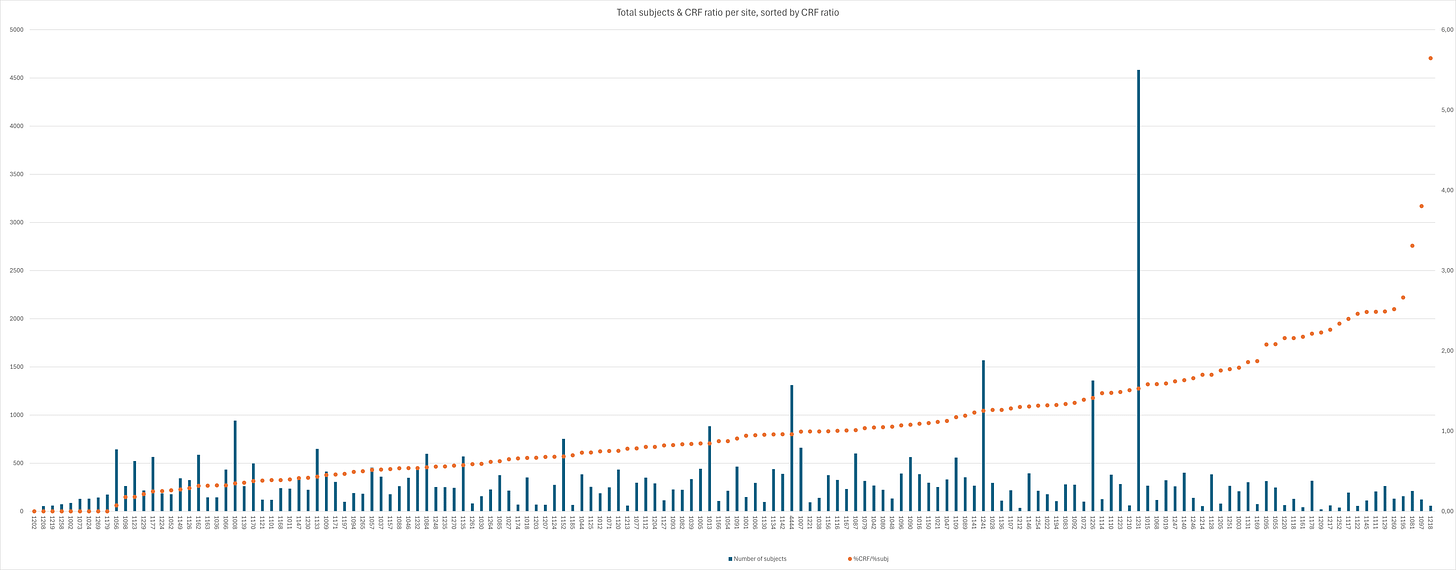TL;DR
This article recounts findings from reviewing Pfizer-BioNTech’s C4591001 clinical trial materials, focussing on manipulations of, and discrepancies between, trial subject source documents and databases. Numerous, serious, individual and systemic data quality issues are identified and examined, with several avenues of further investigation outlined. The reported study data is shown to be an adulterated, incomplete, and unreliable account of a given subject’s health outcomes.
Table of Contents
Introduction- 731 wordsWhich CRFs did Pfizer supply?- 362 wordsDid Pfizer supply all necessary CRFs?- 689 wordsSites- 487 wordsThe Big Fraud- 849 wordsThe “shitshow” CRFs- 1.562 wordsThe study-sized loophole- 1.839 wordsHow to ignore subject reports- 272 wordsAdverse events- 7.024 wordsTrial arm ambiguity- 934 words“Decoy” CRFs- 197 wordsJPG CRF files- 3.222 wordsSuppression of female bleeding events- 1.122 wordsParallel IT systems- 889 wordsEnrolled after 14.11.- 185 wordsMissing nucleocapsid antibody and PCR results- 426 wordsCovid illness data quality- 212 wordsReactogenicity- 1.108 wordsThe protocol amendments- 1.785 wordsDeviations- 1.197 wordsPrimadonna PIs- 1.024 wordsAssorted odds and ends- 500 wordsThe adolescent CRFs- 143 wordsConclusion- 1.551 wordsOverview of the Excel file- 667 words
Introduction
I’ve gone kind of quiet on twitter lately due to this project. End of March I was contacted by HaJo Kremer and asked to proofread a paper on Pfizer-BioNTech’s trial C4591001. An element of his benefit analysis based on the available clinical study reports was that the symptoms associated with covid visits were not reported anywhere. Having followed the PHMPT releases closely, this was the first I had heard of the matter. I began looking into it by reviewing the CO (comments) database for manual entries; this file contains comments associated with blood draws or nasal swabs, and approximately 6.000/149.004 rows refer to samples not collected for a plethora of reasons. The most intriguing comments described covid illness visits being retroactively created at Pfizer’s request.1 This evolved into a concerted reading of the subject source files mid-April. It took me a few days short of a month to amass 170 pages of notes reading these, and I’ve been working on turning all of that into this article ever since.
The enormous amount of material to cover is my excuse for lack of brevity; this is several articles worth of sub-analyses all in one big kahuna. Much of the fraud can only be described by writing a mini-narrative for the subject, and there ended up being quite a few.
It’s also important to emphasize that this is a manual review, thus with a very human error rate. A great example of what this means is JPG CRFs; case reports forms in which single/several pages or even the entire document consists of images instead of searchable text. I found 6 besides the obvious 11 full-image CRFs; when OpenVAET programmatically searched for JPG pages, there turned out to be 68, so what I noticed and wrote down is a lower bound.
Some general considerations: the date format in this article is always DD/MM/YYYY. The timeframe is August 2020 to April 2021, so the month informs in which year the event occurred. Dose 1 & 2 can be either placebo or vaccine depending on the subject, but doses 3 & 4 are always vaccine, as they refer to doses received by unblinded placebo patients. All relevant databases including necessary modifications and subject listings are included in an Excel file2. Where appropriate, footnotes will contain instructions for finding subject groupings. Databases often referred to are CE (clinical events), SV (subject visits), ADAE (analysis dataset adverse events) and ADSYMPT (analysis dataset symptoms). A subject’s ID consists of eight numbers; the first four are the site identifier, and the second four are a sequential enrolment number. Reactogenicity refers to solicited adverse events; these are a list of “expected” adverse events local to the injection site3 or systemic4 in nature, which a subset of trial subjects declared by completing an eDiary.
One thousand and twenty-eight. That’s the number of unique subjects’ Case Report Forms included in Pfizer-BioNTech’s BLA submission for BNT162b2 16+ primary series and released to Aaron Siri’s PHMPT.org . The CRF can be seen as a source document for the databases which are then established; every study procedure, change in status, and health outcome a subject experiences should be recorded in corresponding sections of the form.
A CRF is a sequence of forms, so there’s a medical history form, vaccination visit forms, covid illness visit forms etc. If a subject doesn’t have medical history or didn’t have a potential covid visit, the forms will still be present in the CRF, devoid of data and with “Form Status: NOT STARTED” at the top. The first part of the document is the “final” state of the CRF, showing only the last changes to the data entries. Then come form comments; these are a mixed bag, some files have none, some files have hundreds of pages of “not done” or “not applicable” comments (each comment on a form gets its own page, and some forms have dozens of data entry points), and sometimes even very relevant information not recorded anywhere else. Then come the signatures; the PI has to sign for the CRF being complete, and any edit or change to the CRF invalidates that signature and the PI then has to re-sign it - or not, as is often the case. The second and larger half of a CRF is the audit trail. Here, every change made to the CRF is listed, including queries made by the sponsor.
Which CRFs did Pfizer supply?
The 1028 CRFs represent 2,24% of the 45.748 16+ subjects they claim to have screened. The question why only 2,24% of trial subjects’ source files were turned in is answered in the meeting-correspondence.pdf5 file, which gives us a chronology of the discussion. The first mention of CRFs is made by Pfizer in a communication on 14.9.,6 with their initial offering being only safety narratives for deaths, related serious adverse events, AEs resulting in discontinuation of treatment or study, and all covid cases. On page 498 is the FDA’s answer: “We agree with the proposed plan to include case narratives for all deaths, treatment related SAEs, AEs resulting discontinuation from study treatment and/or study, and that the treatment assignment for the narratives will be unblinded upon successful interim analysis or the final analysis. We may ask for additional safety narratives during the BLA review.”
For the EUA, FDA took exactly what Pfizer gave them and nothing more, a scandalous laxity they corrected for the BLA.7 The BLA CRFs included deaths, SAEs related and not, “anaphylaxis, Bell’s Palsy, lymphadenopathy, appendicitis, and pregnancy exposures and outcomes” due to CBER requests, “AESIs with a Numerical Imbalance that occur at a higher frequency in the vaccine group than the control group”, and severe/repeat covid cases. By no longer requiring all covid cases, they managed to remove most of the positive vaccine arm subject CRFs, and it seems as if CRFs were only submitted for the BLA in May 2021, not for the EUA submission.
“It was worth a try”
The “1.3.2. Sponsor’s Clarification Question to 1.c (sent on 17 March 2021)” is a remarkably bold attempt to reduce the amount of supplied CRFs. Imagine being told “we want these narratives and these CRFs” and instantly respond with layered gaslighting; a) absurd statistical analysis b) “clinical” assessment, the worth of which we’ll be looking at closely.
This section is also important as it reveals Pfizer were unhappy with having to provide CRFs of certain AESIs without applying a filter; this matches the comparatively high rate of anomalies in lymphadenopathy CRFs, and that not all subjects with lymphadenopathy adverse events had their CRFs handed in.
Did Pfizer supply all the necessary CRFs?
Taking into consideration CBER’s requested CRF files, and excluding 12-15 year-old subjects, there are 19 subjects8 who have lymph-related AEs with a physical localisation in the AE description and 1 pregnancy with no CRFs;9 all 833 16+ AEs with a Pfizer serious adverse event number have a CRF.10
With regards to covid visits, the CRFs include 4,56% of total covid visits and 4,48% of convalescent visits,11 and 8,00% of all AEs.12 This is further stratified by toxicity grading.
Grade 4: 100% (167/167)
Grade 3: 54,01% (598/1107)
Grade 2: 9,12% (851/9321)
Grade 1: 4,07% (1062/26046)
2,24% of the CRFs equals a total of 258.499 pages, ranging in length from 106 (Subject 12541006) to 1141 pages (Subject 10521172) with an average of 226 pages for vax trial arm subjects and 274 for placebo.
Much of the pages are forms that were started by mistake, but had to be completed due to the nature of the eCRF program. Especially subjects with multiple covid visits rack up hundreds of pages of “not done” or “not applicable” blood results. Some sites manage this elegantly, others less so, for some there is an obvious learning curve.
The CRFs for sites 1055, 1081, 1096, and 1128 were supplied in bulk files and were amongst the first files released to PHMPT in December 2021 and January 2022, with the last 16+ CRFs included in the third-to-last September 2023 FOIA production. These CRFs have less redactions, except for two files which had invalid redactions: these are supplied separately with the suffix “reissue”.
There are CRFs in the Pfizer-BioNTech 12-15 PHMPT production, which is still ongoing has apparently concluded13; these subjects are included in the 16+ datasets available but their CRFs are only in the 12-15 documents and will be covered separately. This is also why this age group is regularly excluded in this article.
The CRFs comprise:
482 BNT subjects
43 with only one dose
541 placebo subjects
47 with only one dose, of which 16 and 4 received 1 and 2 doses respectively after unblinding despite being previously discontinued
104 with only two doses (no unblinding doses)
89 with three doses, 74 of which received dose 3 within 21 days of data cutoff
30114 with four doses
5 subjects discontinued before dose 1 without trial arm assignment
all male, 24, 26, 32, 38 and 38 years old.15
Arranged differently, 151 CRF placebo patients never received a dose of BNT162b2, all other 877 CRF subjects received at least 1 dose of vaccine. It’s surprising that more subjects discontinued after dose 1 in the placebo arm than in the BNT arm.
Arranged by reason for inclusion, the CRFs break down as follows. Note: the dose designations for placebo subjects refer to the last dose recorded for that subject, which is not necessarily the dose the SAE ocurred at.
Exposure during pregnancy 100 CRFs: 44 BNT 56 Placebo (dose 1: 6, dose 2: 28, dose 3: 7, dose 4: 12)
Lymphadenopathy 178 CRFs: 92 BNT 86 placebo (dose 3: 8, dose 4: 78)
Covid16 38 CRFs: 1 BNT, 37 placebo (dose 1: 5, dose 2: 11, dose 3: 10, dose 4: 11)
Cardiac17 107 CRFs: 51 BNT 56 placebo (dose 1: 8, dose 2: 10, dose 3: 8, dose 4: 30)
Lithiasis 18 44 CRFs: 28 BNT 16 placebo (dose 2: 1, dose 3: 3, dose 4: 12)
Clots and bleeds 19 68 CRFs: 33 BNT, 35 placebo (dose 1: 2, dose 2: 5, dose 3: 4, dose 4: 24)
Cancer 92 CRFs: 49 BNT, 43 placebo (dose 2: 15, dose 3: 5, dose 4: 23)
Infections and respiratory20 78 CRFs: 35 BNT, 41 placebo (dose 2: 8, dose 3: 11, dose 4: 22)
Allergy/anaphylaxis 20 CRFs: 6 BNT, 14 placebo (dose 1: 4, dose 3: 4, dose 4: 6)
Bowel21 80 CRFs: 42 BNT, 38 placebo (dose 1: 1, dose 2: 3, dose 3: 8, dose 4: 26)
“Osteo-”22 36 CRFs: 16 BNT, 20 placebo (dose 2: 3, dose 3: 3, dose 4: 14)
Drug/psychiatric 22 CRFs: 7 BNT, 15 placebo (dose 1: 1, dose 2: 6, dose 3: 4, dose 4: 4)
Sites
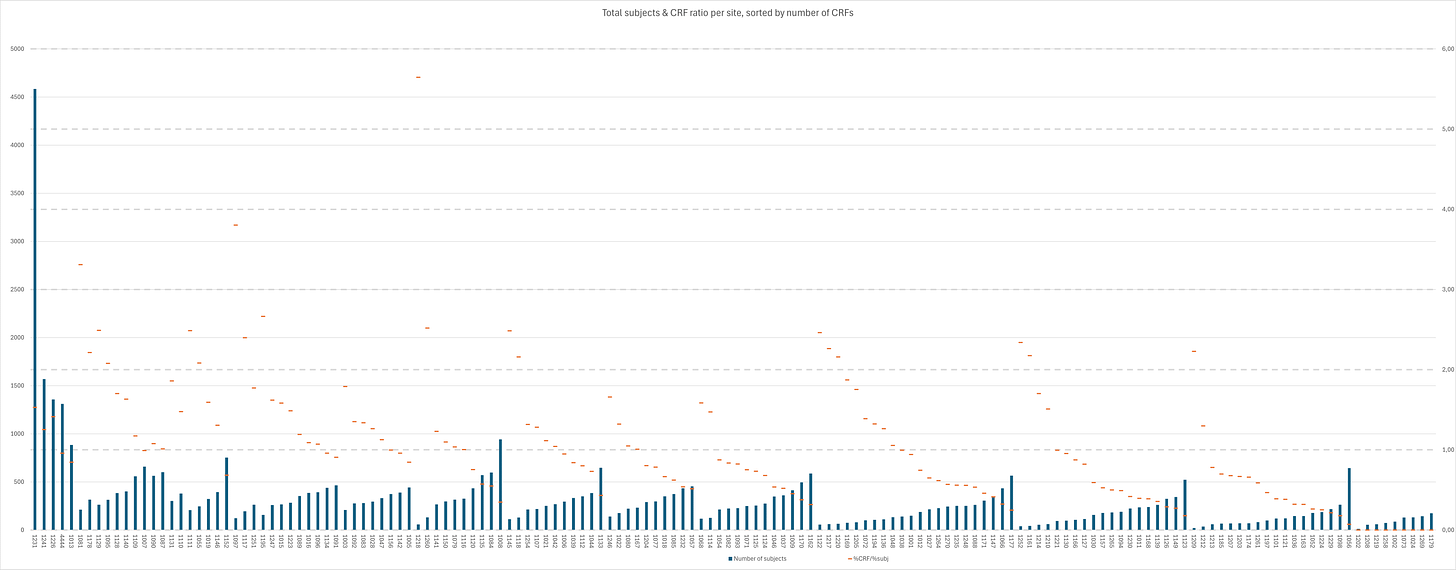
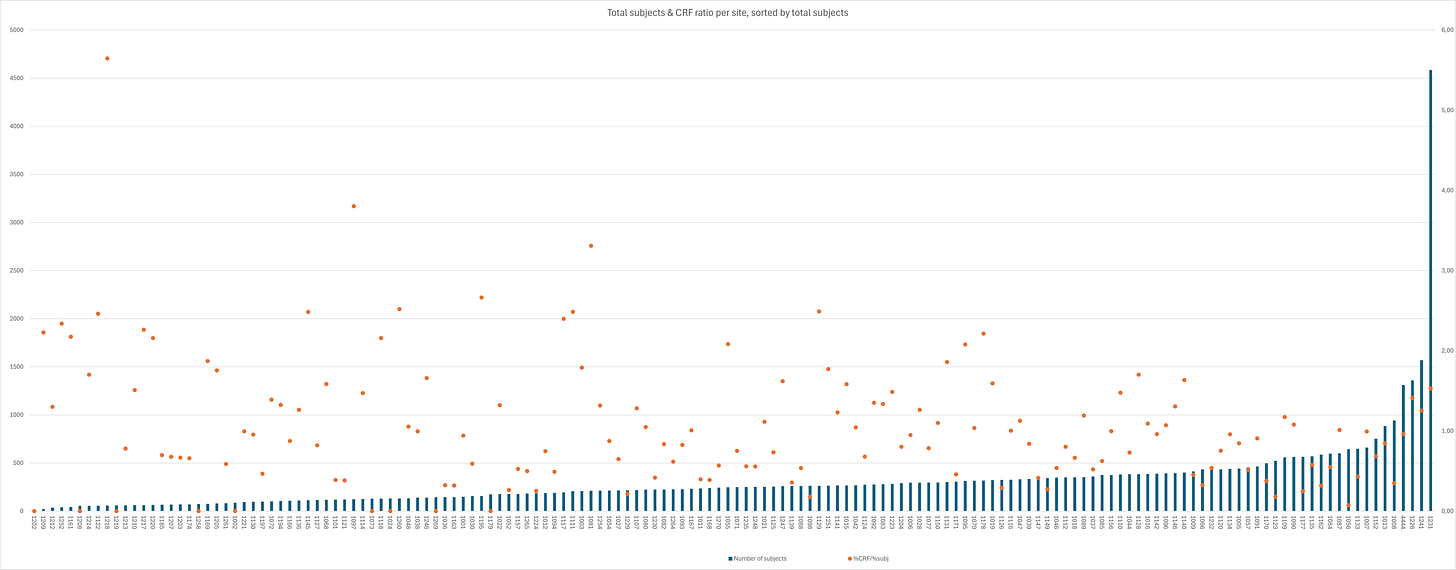
When taking a look at the distribution amongst sites, it quickly becomes clear there are quite significant differences. By dividing the % of total CRFs by the % of total subjects for each site, we get a “sanity check” ratio - any site <1 has less CRF files than it should. Site 1218 had only 58 subjects screened, but supplied 7 CRFs, giving it the highest CRF/subject rate amongst sites of 12,07%. On the other end of the spectrum are eight sites with a total of 866 subjects with no CRFs23 and then Site 1056; 1 CRF for 643 subjects. A total of 50 sites24 with 14.434 subjects have a ratio < 0.6, and a total of 38 sites25 with 12.432 subjects have a ratio >1.4. The 19 sites with a ratio >2 together only have 2.758 subjects and 149 CRFs, corresponding to 5,53% of subjects yet 14,49% of CRFs; conversely, the 37 sites with a ratio < 0.5 have 10.061 subjects and 63 CRFs, corresponding to 20,8% and 6,13% of their respective totals.
The inorganic CRF distribution amongst sites is emphasized further when regarding the distribution of CRF within sites.
Site 1178 is an excellent example: 317 subjects, 15 CRFs, very high %CRF/%subj ratio of 2.21. The subjects with CRF files are 1012, 1015, 1025, 1048, 1061, 1073, 1107, 1122, 1138, 1164, 1167, 1257, 1287, 1293, 1300. The gap between 1167 and 1257 is slightly auspicious, but otherwise this is a plausible distribution, especially regarding the actual contents: 5 cases of kidney or gallbladder stones, 5 cases of lymphadenopathy, three cardiac cases, one bowel obstruction, one exposure during pregnancy, along with extraneous AEs including two counts of cancer, one pre- and one post-vaccine receipt.
Going down the listed sorted by ratio, Site 1079 has 316 subjects yet only 7 CRFs, giving it a ratio of 1.04, still above average. CRF subjects are 1004, 1044, 1076, 1183, 1199, 1228, 1246, with two cancers, two strokes, one central retinal artery occlusion (in a placebo patient), one total knee replacement, and one case of seizures; no incidence of lymphadenopathy, nephro-/cholelithiasis or pregnancy.
At the “shallower” end of the pool, there are sites like 1126, 324 subjects, 2 CRFs, ratio of 0.29. One allergic reaction, one case of lymphadenopathy, both in subjects who first received placebo. It should be noted that this site was run by Kaiser Permanente Sacramento & Santa Clara.
While this can be solidified mathematically I’m sure, it’s enough for me to entertain several options:
different sites got different or deactivated product
PI honesty/site diligence, or lack thereof
Synthetic data
Extensive use of the medical history loophole
Selective instruction on use of the eDiary apps
Let me know in the comments if you can come up with reasons for these massive discrepancies in CRF coverage.
The Big Fraud
Imagine, if you will, a clinical trial protocol so counterintuitive and arcane in its adverse event reporting structure that it has to be explained repeatedly to each expert investigator and trial site. Here is the twist:26
Potential COVID-19 illnesses that are consistent with the clinical endpoint definition should not be recorded as AEs.This means there are two pools; one is adverse events in terms of safety, and the other is “potential covid illnesses” in terms of “efficacy”. The only place where the symptoms of potential covid illnesses are reported are in the covid-positive narratives. None of the other narrative groups (like pregnancies, deaths, AESIs etc.) report on potential covid illnesses or symptoms experienced in connection with them. All other narratives report only the contents of ADAE, the “adverse events” pool - even if there are months of diarrhea in a potential covid illness. The reactogenicity a subject experienced is not reported in the narratives either, adding a third “pool” for the ~12k subjects in the solicited adverse events subset. There are also two impactful protocol design elements at play here: the overlap between reactogenic solicited adverse events and covid symptoms,27 and an embargo on swabs seven days after each dose. What this means in practice? An example:
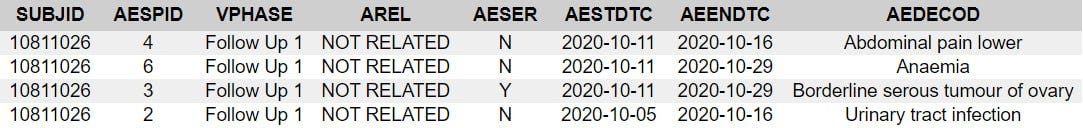
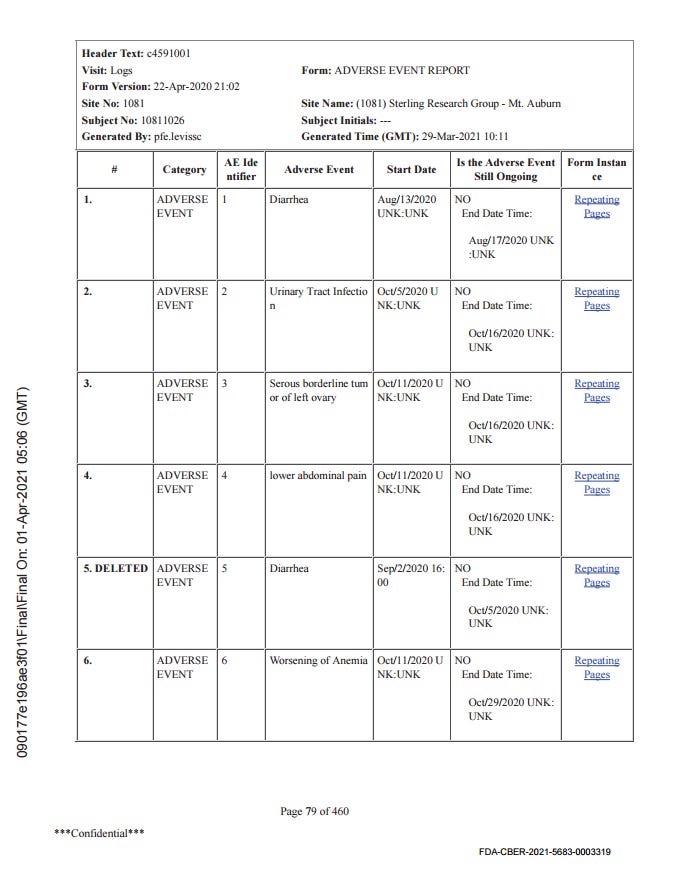
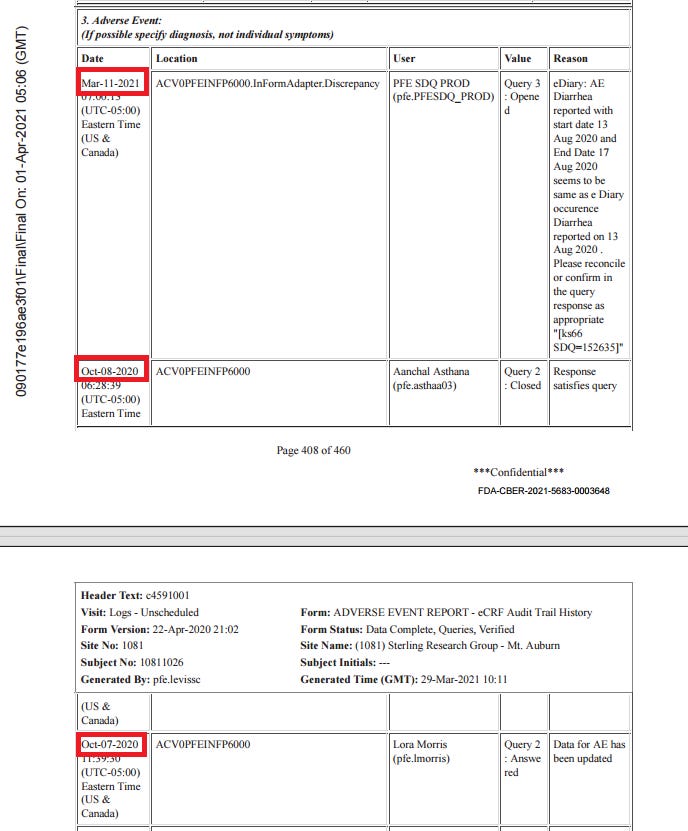
Subject 10811026
67 year old woman, BMI of 41.9, 20 medical history items related to obesity and aging, b2 reactogenicity subset assignment, dosed 12.8. and 2.9. Her CRF is included due to the SAE of “serous borderline tumor of left ovary”. This is the subject narrative (no prose); note the far-left column “AE Number” from 1 to 4.
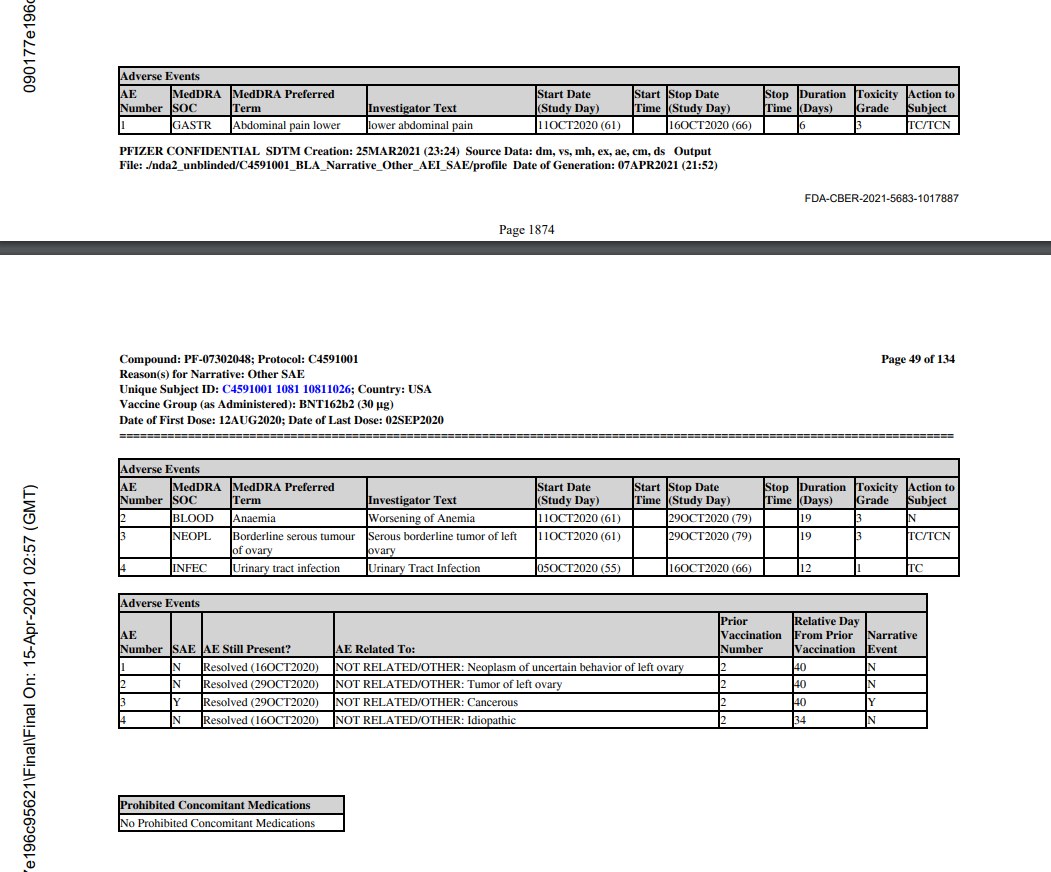
Next come the entries in the adverse events analysis dataset, ADAE.
The missing numbers in the AESPID column represent deleted adverse events, as can be seen in the CRF.
Essentially, AESPID is the CRF-specific AE identifier, AESEQ the identifier in the database.
has exploited this to find gaps in the AESPID column and thus deleted adverse events in points 61-66 of his most recent paper.Your eyebrows might already be climbing towards your hairline, but there are more pieces to assemble yet. Next are the entries in ADSYMPT (potential covid illnesses) and CE (clinical events).
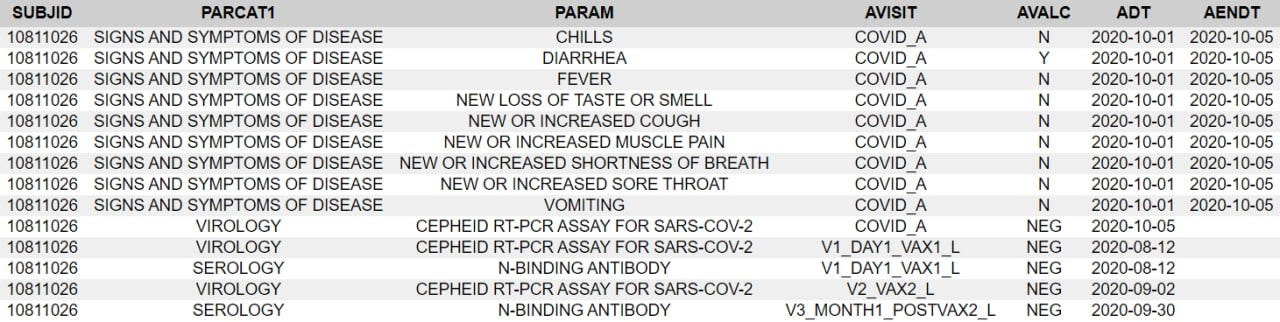
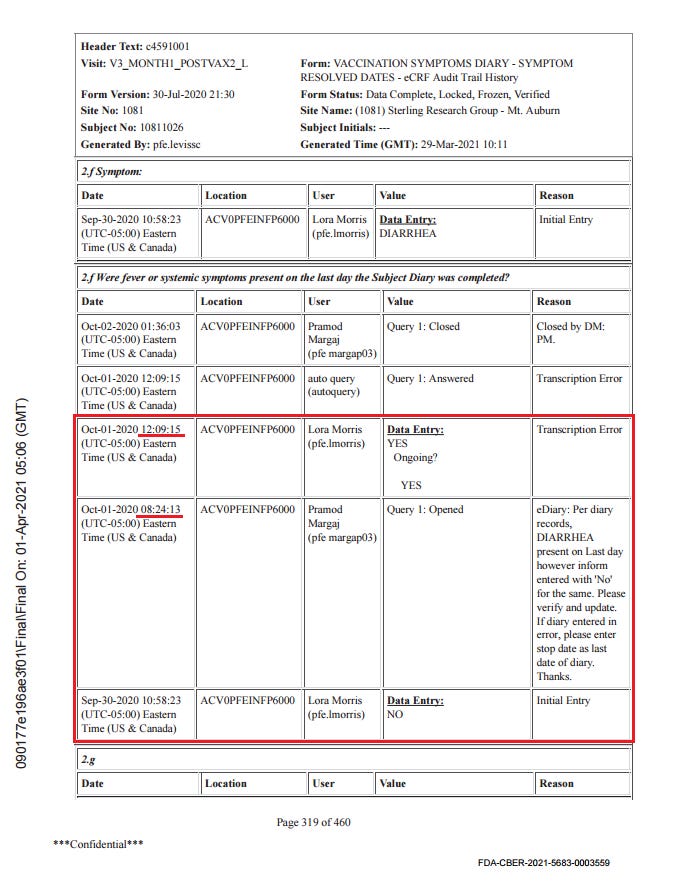
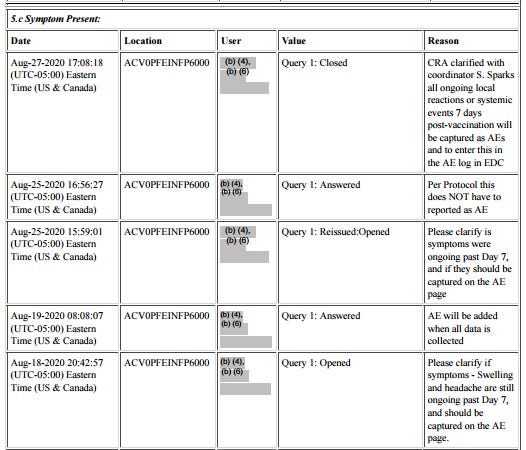
The subject experienced diarrhea twice, for four days after dose 1 and for over a month after dose 2. Here is the order of events according to the CRF audit trail: visit 3, the one-month post-dose 2 visit, occurred on 30.9. The site made an error in transcribing the reactogenicity eDiary data by entering “no” instead of ongoing, and were queried on this by Pfizer at 8:24 on 1.10., changing the entry at 12:09.
Then there was presumably a telehealth telephone contact because the first entry to the AE log occurs at 14:09. It takes a full five months28 for someone on Pfizer’s end to catch on that the diarrhea was already captured in the reactogenicity eDiary, but the site must not have gotten around to changing it, because it’s present in the CRF as AESPID #1, but deleted in ADAE.
The diarrhea AESPID #1 was initially entered on October 1st as lasting from 3.9. > ongoing, changed to 13.8.-17.8. on October 7th, so they changed the instance of diarrhea the AE refers to. Seeing as the first instance was a reactogenicity eDiary entry, it shouldn’t have been entered anyway. In response to a query that the diarrhea doesn’t have an end date, the site explicitly replies “The ongoing diarrhea is captured in the COVID Illness pages”.
The covid illness visit the diarrhea was moved to was only created on October 5, which is also the date of the corresponding central lab swab. Fully in line with protocol, as a swab has to be within 4 days of symptoms ending, and entirely nonsensical clinically. On October 7th AESPID #1 was edited to refer to the initial bout of diarrhea, which was then post-facto deleted by Pfizer. The deleted diarrhea AESPID #5 referring to the second instance of diarrhea was created on November 3rd and deleted on November 5th, after the following Pfizer clinical query: “CLINQUERY: Diarrhea of 02Sep-05Oct20 appears to be captured in the COV ILL CRFs. If this is the same event, please capture only in the COV ILL SOD and not as duplicate in the AE listing.”
As a result, the considerable health event of more than five weeks of diarrhea in a ten week timespan is not reported as an adverse event for this subject anywhere except in the database, even though it being recorded as an ongoing dose 2 reactogenicity eDiary event, which according to protocol mandates AE reporting anyway.
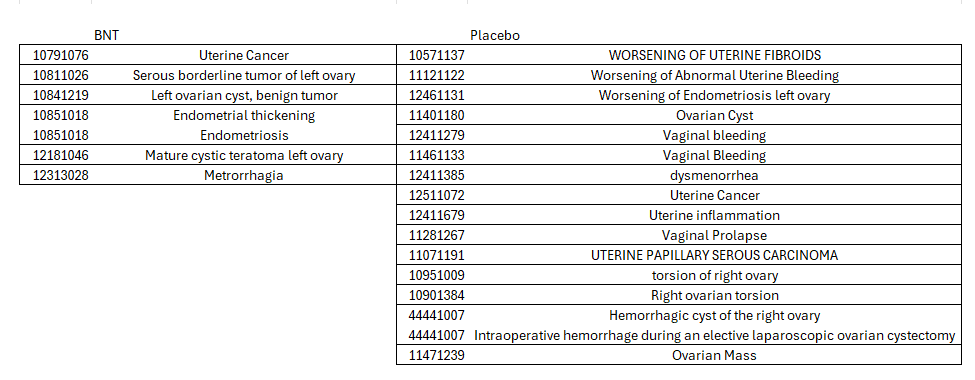
This CRF is also highly suspicious of being an unreported female vaginal bleeding case. How else can a “worsening of anemia” be related to an ovary tumor, if not for bleeding? Yet the specific term is stringently avoided, even including reference to the “abdominal pain, lower” adverse event: “CLINICAL SAE report reports subject had Abdominal Pain from Oct 11 to 16, 2020. Please assess for safety impact, and update with an event for this complaint if appropriate”.
The “shitshow” CRFs
10811026 has a bit of everything so it’s a great example, but there are a couple of CRFs that are especially noteworthy due to the extent of Pfizer intervention, site errors and the effects of both when trying to execute an intentionally obtuse protocol.
10911197 - 54 / M / placebo / BMI 25.8 / doses: 28.8. + 17.9. + 1.3.
This subject’s cocaine habit kept everyone on their toes. Subject had covid illnesses from 3.-15.9., 26-30.9., 28.10.-12.11., and 16.11.-2.12., and has unacknowledged covid symptoms on 6. & 11.10, and on 2.2. The second covid illness visit occurs 19 days after end of that illnesses’ symptoms and overlaps with visit 3; similarly, the fourth covid visit is entered over a month after the fact, in January. The adverse event of diarrhea starts directly after the first covid illness and overlaps the second, but these illnesses don’t have diarrhea as a symptom. Similarly, there is an adverse event of “nasal congestion” for a week in October in between the second and third covid illnesses, which should be a covid illness too, according to protocol. The toxicity grade 4 “poisoning by cocaine” started out as an opioid overdose.
11291183 - 61 / M / placebo / BMI 28.9 / doses: 15.9. + 6.10. + 25.1 + 15.2
This subject starts off the trial on the decidedly wrong foot, as dose 1 is delayed due to “recent non-study vaccination” with a flu jab on 1.9. - by one day, to the 15.9. The first potential covid illness visit on 19.9. is marked erroneous and is named covid_b. The second covid illness, covid_a, is from 28.9. to 3.10. with the illness visit on the 2.10, the diagnosis of “gastroenteritis” is dated 6.10, and includes the extra symptom of abdominal discomfort, corresponding to the deleted adverse event of “acute gastroenteritis”, entered on 10.11. and deleted on 12.11. Also deleted is the adverse event of bilateral leg muscle pain from 17.-19.9., which was “moved” into the deleted first covid illness covid_b. As it turns out, this first “covid illness” was created on 14.10. due to Pfizer’s illness visit query bullying, and deleted on 24.11.; the second covid illness, covid_a was entered first, on 2.10. The bilateral leg muscle pain never makes it back onto the adverse event log, which is especially irregular due to “left shoulder soreness” remaining coded for dose 1, and a week of bilateral upper extremity pain and bilateral shoulder pain after dose 2 being coded with no objections.
11311204 - 84 / M / placebo / BMI 30.5 / doses 22.9 + 15.10 + 21.1
This subject died of “worsening aortic stenosis” and cardiopulmonary arrest after dose 3. A primary care provider visit which occurred during covid_c was erroneously also entered for covid_a, but nobody noticed; furthermore, covid_c has no end date until it’s changed to 15.2., the day of death; however, as it started before dose 3, the data now suggests Pfizer jabbed a symptomatic patient and he subsequently died: "Subject has passed away. End date updated to date of death. Shortness of breath was still ongoing up until the time of death” This especially perplexing as there was a convalescent visit for covid_c on 4.1. The database also includes a medical history item of aortic stenosis ongoing since 2010 which is not present in the CRF.
11401009 - 40 / M / b2 / BMI 30.2 / doses 31.7 + 20.8
After dose 1, the adverse events of injection site pain and injection site swelling are entered into the adverse event log, despite the subject being part of the reactogenicity subset. This mistake is bizarrely repeated and done wrong even worse for dose 2: the reactogenicity event of fever is entered as an adverse event again, but this time it’s not only deleted, but ALSO recorded in a covid visit four days longer than the deleted adverse event.
11661047 - 50 / M / placebo / BMI 19.3 / dose: 31.8.
Received only one dose of placebo due to the adverse event of “dizziness” on day one. There is bizarrely also a covid visit on the day of dose 2 (22.9.) describing the additional symptoms of fever and shortness of breath for one day on 31.8.:“[Specimen was taken IN-CLINIC because this started as a regular Visit #2; per CRF/CRA instructions, all available data was used to create an illness visit instead.]” with a diagnosis of “[Allergic reaction to vaccine #1], entered retroactively on 7.10. The adverse event log contained fever and SoB with AESPIDs 1 & 2 from 24.9. until 14.10 and 13.10. respectively, until the site succumbed to Pfizer queries and deleted them. A form comment on 28.9. states “Unable to remove date (required per query: remove V2_VAX2 data because vaccine was withheld by investigator).”, but on the very next page, a form comment from 6.11. has a different view of events: “Patient did not receive vaccine #2 per patient's own choice and d/c'd study. Study procedures do not apply”. Another form comment dated 10.11. and deleted 17.11. states: “N/A pt discontinued; meant to enter CONVA_A visit.” Notably, the subject was only discontinued from treatment, not from the study, as evidenced by a covid illness in December. There are also a bunch of form comments entered on 26.12. with the following absurd text: “PROTOCOL ONLY REQUIRES STANDARD OF CARE PROCEDURES. THIS SITE DOES NOT CONSIDER THIS TO BE SOC - THUS NOT REQUIRED, PER PROTOCOL. THUS NOT DONE, YET N/A IS MORE APPLICABLE BECAUSE NOT REQUIRED (I.E., NOT SKIPPED/MISSED AS IN "ND").” on the non-essential covid_a forms such as oxygen therapy, severe illness details, local laboratory data etc. The same occurs with covid_b (10.-14.11.) and covid_c (19.-21.12.), the form comments dated 11.1. and 8.1. respectively, so covid_c’s form comments were entered three days prior to covid_b’s. Another important point is that in the CRF, covid_c is marked “ongoing? yes” on 8.1. when it’s entered, yet in the database there is an end date. The “standard of care” message occurs 355 times throughout the CRF, and each instance generates it’s own page. There is a “subject safety-related” unblinding entered 12.10. and deleted one minute later. Remarkably, despite ReVax unblinding contact on 18.2., there is no dose 3 listed and no Pfizer query related to it’s absence.
12311352 - 45 / F / placebo / BMI 33.3 / doses: 15.8 + 4.9 +10.3.
The notes for this subject take up an entire page:
The insanity of the covid illness/adverse event reporting rules leads to remarkable results for this subject. The adverse event log has an event of vomiting from 29.8.-1.9., but the “potential covid illness symptoms” show vomiting as a symptom from 20.8. - 7.9. and from 14.-19.1. The subject received dose 2 while in a “covid illness” and while experiencing an adverse event of UTI.
12313653 - 54 / F / placebo / BMI 30.9 / doses: 24.8 + 13.9 + 25.2
This subject has a three-month covid illness with the diagnosis “ischemic stroke” lasting from 10.9.-7.12. The adverse event of stroke however is from 6.11.-22.1. The CRF contains very entertaining arguments between Pfizer and the site, regarding for example the inclusion of “significant neurological dysfunction” due to ischemic stroke in the “severe covid illness” form despite there a) being no positive swab and b) stroke already being logged as an adverse event. The subject received dose 2 six days after covid_b began. There is also an adverse event of “Exacerbation of bronchial asthma, dyspnea”, related to “Baseline bronchial asthma”, dated 15.9 and described as ongoing on 15.10., the day it was entered. It was deleted five days later because “This adverse event is dismissed, the Covid-19 Disease will be opened to the volunteer.”, which was sufficiently indecipherable for the adverse event to be undeleted briefly six days later in order for someone with better English skills to reformulate: “The cross out is removed to answer the query. This adverse event is crossed out because it is not considered an adverse event but rather a probable covid disease”. The subject also experienced an asthmatic crisis from 1.3.-13.3. after receiving dose 3 on 25.2., which they didn’t have to delete from the adverse event log because it was only ever entered on the covid illness form.
12481120 - 74 / F / placebo / BMI 36.8 / doses: 11.9 + 21.10 + 15.1 + 5.2
The subject has an adverse event of “mild stroke” in the CRF, yet the database lists “acute heart failure”. Acute heart failure is mentioned nowhere in the CRF at any point in time. The adverse event has a start date of 23.9, twelve days after dose 1. The initial entry of “ongoing? yes” is made on 7.10 and never re-queried or changed. The subject also tested positive while in hospital, yet denied having any protocol-defined covid symptoms and was thus an “asymptomatic” case, which allowed receipt of dose 2 on 21.10, and further doses after unblinding. It is highly anomalous that this first “asymptomatic” covid illness has a toxicity grade of 0 entered in the CRF, yet no corresponding entry in the Clinical Events database, which only lists the second potential covid illness, which was initially an adverse event of diarrhea for <24hrs six days after first unblinding dose, and was deleted one month after entry. ADSYMPT (analysis dataset symptoms) lists both potential covid illnesses.
The study-sized loophole
Abuse of “potential covid illnesses” to keep health outcomes off the adverse event report form was commonplace; constantly coaxed, coached and queried by Pfizer employees. 374/1028 CRFs involve at least one covid illness, and 140 of these are highly suspicious of being fraudulent29. On the other hand, this resulted in what effectively are adverse events being reported beyond the Visit 3 “one-month-post-dose-2” limit, and potential covid illnesses had very high CRF completion requirements when compared to adverse events, for example lab values, oxygen therapy details, imaging results etc.
Here are typical covid illness-related irregularities:
10011093 - 59 / M / b2 / BMI 28.2 / doses: 29.7 + 19.8
In the adverse event audit of “chest pain” on page 401 that was first coded “angina” and then “left heart catheterization”, Pfizer asks on 18.1. whether a covid visit was conducted for the symptoms of chills and dyspnoea. On page 322 and following, a covid illness with visit date 13.1. and a central lab self-swab dated 21.1. are entered on 26.1. Especially remarkable as the covid illness doesn’t have an end date in the CRF, but does in ADSYMPT, despite the CRF being edited as late as Visit 4, recorded with entries from 9.3.2021.
10051293 - 60 / M / b2 / BMI 27.8 / dose: 19.10
In the adverse event audit of acute respiratory failure in the context of supposed cocaine overdose with cardiac arrest and resuscitation on 8.11. on page 310, Pfizer demands a covid visit be done on 13.11. On 16.11., the site follows suit, with an entry on page 204 dated 19.11.: “COVID illness visit completed off of hospital medical records - no symptoms listed on medical records - covid visit completed per sponsor request.”
10121163 - 40 / F / b2 / BMI 20.5 / dose: 9.9
In the comment section on page 100, site staffer Sahara Vega writes on 5.11.: “Visit entry being done to capture shortness of breath reported during a phone call on 9/16/2020. Not an actual possible covid illness visit.” Indeed there is no swab associated with the visit in ADSYMPT or MB.
10161289 - 17 / M / b2 / BMI 22.4 / doses: 17.8 + 9.9 + 20.1 + 12.2
The covid illness from 7.-11.10., starting day of dose 2, includes the extra “other” symptom chest tightness on page 270, which is reflected nowhere in the corresponding narrative nor anywhere in the database.
11111103 - 66 / M / b2 / BMI 32.6 / doses: 11.8 + 31.8
The covid illness from 13.9. > ongoing was entered on 23.9. On page 601, the “other” symptom of anemia is added to the covid illness, and remains the only mention of this adverse event until the AE log on page 873 is created on 10.3.2021. The anemia adverse event entry also begins a month later than the covid illness. Remarkably, on page 582 the site confirms the covid illness to be ongoing on 24.2. with no end date noted in the CRF, yet there is an end date of 27.2. in ADSYMPT. Nonetheless, 167 days earns this subject the dubious title of longest potential covid illness with an end date; podium spots 2 & 3 go to 11681007 with 145 days and 10071115 with 139 days.
12261210 - 30 / F / b2 / BMI 20.1 / doses: 17.8 + 8.9
In the covid illness from 6.-8.12 on page 255, chest pain is listed as an “other” symptom. There is no other record of this health event.
10061094 - 35 / F / b2 / BMI 20.3 / doses: 24.8 + 18.9
The adverse event of sinusitis is entered on 18.9., recoded to upper respiratory tract infection on 22.9., and deleted on 23.10. with the reason “CAPTURED AS COVID_A” on page 213. The site has a bizarre reponse to Pfizer’s queries for testing on 15.10., page 218: “AE occurred prior to protocol amendment 6 approval and irrespective of perceived etiology, was not included in protocol amendment 5. Investigator deemed AE not a COVID-like illness.” Cov_a is then finally entered on 21.10. p186, and the AE deleted two days later. Page 199 reveals that the Visit 2 swab was deleted and moved into the covid visit on 21.10. Remarkably, on page 152 “recurrent sinusitis” is added to the medical history on 21.10., despite already being recoded to “upper respiratory infection”. The covid illness also includes “discolored mucus” as an “other” symptom.
10441194 - 56 / F / placebo / BMI 23.1 / doses: 18.10 + 6.11 + 5.2 + 26.2
An adverse event of ataxia is entered on 10.3. at 14:42, then a covid illness starting 7.3. with the “other” symptom of ataxia is entered at 16:35. At 13:36 on 11.3., the adverse event of ataxia is deleted following a remarkable Pfizer query at 11:24 on page 648: “CLINQUERY: Potential COVID-19 listed symptom ATAXIA is already documented under the COVID Illness SOD CRF. PP Section 8.37, events consistent with the clinical endpoint definition are not to then also be recorded as AEs. Update AE CRF as appropriate.” This subject has unlogged adverse events on page 617 of opioid overdose after developing breakthrough cervical radiculopathy pain. Despite it being a stable condition at enrollment and the new additional medication causing side effects, some of which are even listed as adverse events, the site refrained from listing this as an SAE.
A troublesome observation to be made here is the abuse of “fatigue”. It is both a solicited systemic adverse event and a potential covid illness symptom, and this abuse in the sense of “asthenic condition” or weakness dilutes the spectrum of ailments it could stand in for, to include things like Guillain-Barre, or in this case opioid OD.
11781061 - 38 / M / b2 / BMI 24.7 / doses: 1.9 + 14.10
On page 99-100, the AE log shows that 5/11 adverse events were deleted. Covid illness B where they were moved to was created on 5.11., with start date 14.10., same date as the delayed second vaccination. On page 390 the symptoms are confirmed to be ongoing as of 23.2.: “Coordinator spoke with subject and subject states continuing symptoms of daily headaches and nausea. Subject reports that he is scheduled to see his primary care physician.” The subject’s second dose was delayed from 22.9. to 14.10. due to corticosteroid use (page 316), but there is no corresponding adverse event logged, nor a concomitant medication form filled out. A site response on page 311 dated 28.9. states: “Please reference other queries regarding this visit. Subject met delay criteria and visit will not be completed until October.”; there are no other queries to this visit to reference I could find. Covid_A is from 2.-11.10. so that only began after the subject was already out of window for his second dose. Pfizer querying why a second covid visit was started within four days of symptoms on page 389 and then closing the issue after an opaque allusion to “additional discussions” is suggestive of extensive off-the-record sponsor-site interaction. This subject’s headache and nausea, validated as ongoing for more than four months, were deleted from the AE log twenty days after the adverse events began, never to return.
12311118 - 31 / M / b2 / BMI 36.6 / doses: 13.8 + 4.9
Subject has three covid illness visits but should actually have five. Covid illness A was extended from stop date 26.8. to stop date 23.9. on 27.10 on page 350, following a subject eDiary report of covid symptoms on 19.9., page 327. Covid illness C was changed from 21.12.-2.1., to erroneous visit on 20.1., then to 27.1.(entered 2.3.)-6.3.(entered 9.3.). The initial covid_c visit was rolled into covid_b, despite there being local and central lab swabs, which were deleted in the merger.
12313674 - 58 / F / b2 / BMI 23.7 / doses: 24.8 + 13.9
Covid illness A on page 246 had a symptom stop date of 16.10. entered on 2.11., which was changed to “ongoing? yes” on 8.11., with a final symptom stop date of 16.11. entered on 30.11. The fraudulent extension of this covid illness is further supported by the subject’s eDiary report of covid symptoms on 2.11. which the site did not respond to until Pfizer queried the absence of an initiated covid visit on 8.11, page 230.
There are even subjects where the covid illness system was abused without there being a covid visit in the database:
10391010 - 84 / M / b2 / BMI 24.6 / doses: 21.8 + 9.9
A covid visit with a self-swab completed on 10.9. (page 341) is deleted one week after entry. An adverse event of fever for 12 hours on 10.9. is entered on 17.9. (page 447), the same day the covid visit is deleted. This subject died on 18.11. and the site reported being informed on 9.12.
10891182 - 47 / M / b2 / BMI 23.5 / dose: 25.8
A covid visit was created retroactively from medical records due to a Pfizer query, as described in a comment on page 170. The covid visit is created on 9.10., following Pfizer queries on the adverse event of “viral illness” 8.-9.10. page 506, and even includes a local swab dated 27.9. The covid visit is populated with data on 12.10., and the adverse events of viral illness and vomiting are deleted on the 16.10. (page 475). The covid visit is then marked “erroneous” and all data set to “not done”(p396) on 20.10., but the adverse events never make it back into the log.
11171058 - 34 / M / b2 / BMI 24.2 / doses: 25.8 + 15.9
On 21.9., a covid visit is created retroactively with the symptom of muscle pain from 26.-28.8., and the symptom of fever is changed to “present: yes” on 13.10. After the investigator deems the event to be reactogenicity, all data in the covid visit is purged starting 25.11.(“EVENT BEING REMOVED FROM SYSTEM” p341), neither symptom is added to the AE log. This subject also had an adverse event of myalgia after dose 2 besides the lymphadenopathy (unrelated) and headache (related) that are listed in the database; however, on page 306 a redacted user enters the following on 19.10.: “SEE SOURCE AE FORMS 2,3,4. PER INVESTIGATOR PROGRESS NOTES, NO SUSPICION OF COVID ILLNESS PRESENT AT THIS TIME. v2 ON 15SEPT2020, SYMPTOMS START ON 16SEPT2020. REACTINOGENIVITY PERIOD. THIS EVENT SHOULD BE REMOVED.” However, the adverse event of myalgia, initially entered on 17.9., had already been deleted after a Pfizer query dated 4.10. stating: ”Clinical Query: Unfreezed now. As myalgia is recorded on the SOD CRF as a potential COVID-19 symptom it should not also be recorded on the AE CRF. Thanks.” on page 482. As this subject was not in the reactogenicity subset, this manipulation of the record over months resulting in 3/5 adverse events entirely unreported and ultimately only visible due to the subject’s lymphadenopathy requiring CRF submission, casts considerable doubt on Site 1117’s interpretation of the protocol and Pfizer’s supervision of the trial.
How to ignore subject reports
In a trial with such a focus on efficacy that adverse event reporting was inherently hamstrung, it was surprising to discover an established methodology for a subject-reported symptom to be ignored and no covid illness visit to result: all the site had to do was nothing, and when Pfizer queried why there was no covid visit, state that symptoms were not related, not clinically significant, or that the subject made the eDiary entry in error.
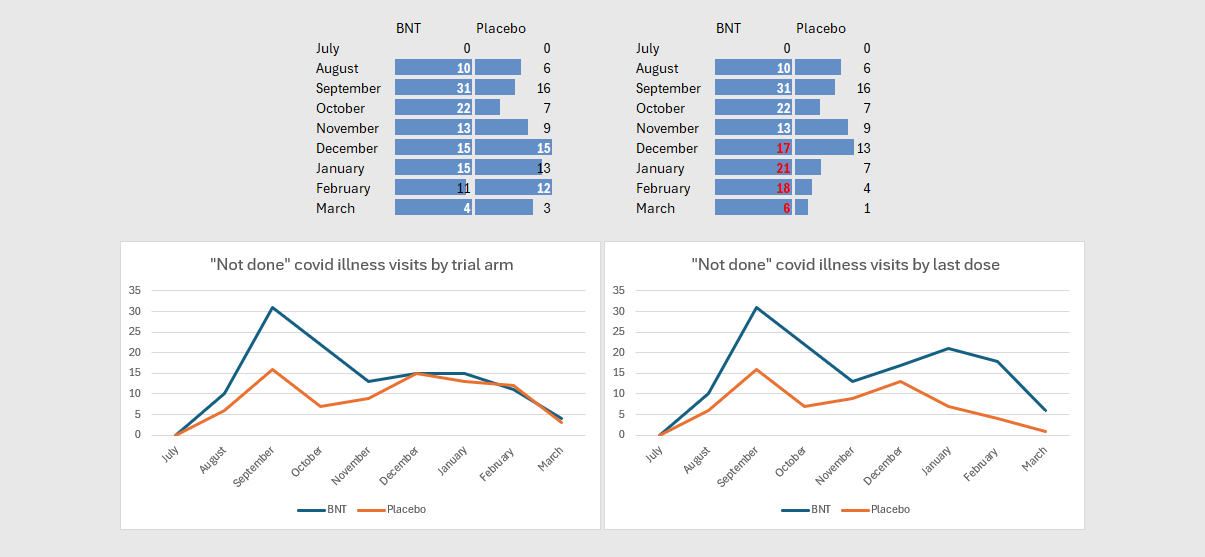
153 CRFs have between one and five “not done” covid visits each; 92 in the BNT arm30 with 120 missed visits and 6131 placebo subjects with 80 missed visits in total; 1332 placebo subjects have their first missed visits after unblinding. Of the 92 BNT subjects, 51 (55%) do not have a covid visit, corresponding to 32 placebo subjects (52%).
38 BNT and 27 placebo subjects amongst the 153 with missed/not done covid visits have no related deviations33, which includes 29 and 19 BNT/placebo subjects respectively who also don’t have a covid visit. A software-based approach to these CRFs might well yield more.
To recap briefly: under the guise of separating “efficacy” from “safety” data and avoiding record duplication, Pfizer designated all potential covid illnesses as “expected clinical endpoints” which are not to be reported as adverse events, thereby creating an unreported pool of health outcomes, as “symptoms” experienced during a “potential covid illness” without a positive central lab PCR are described nowhere. The protocol proved to be so fundamentally counterintuitive that there is barely a single CRF-documented covid illness visit without Pfizer intervention, with 151/374 CRFs involving a covid visit crossing the threshold into potential abuse.
Adverse events
Anything you can think of to falsify reporting of adverse events was done in this trial: from super soft coding (“chest pain” with surgical intervention, “exacerbation of osteoarthritis” meaning bilateral total knee replacement) over straight up lies and omissions, to enormous delays in reporting and even not reporting in the CRF at all.
Before diving into the data, there are two important protocol design choices to consider besides the “potential covid illness” loophole already covered; the medical history loophole and the Visit 3 reporting timeframe.
If an adverse event occurred during the trial and was also present in that subjects’ medical history, it was only to be recorded as an adverse event if, in the investigator’s opinion, it represented a worsening or exacerbation.
After Visit 3, one month after the second dose, only serious adverse events were to be reported, and concomitant medication documentation became much looser, as immunosuppressant and corticosteroid use was no longer required reporting.
Another important point is the Pfizer safety database. SAEs were to be faxed (!) to Pfizer, and an extremely frequent occurence34 in the CRFs are reminders to include an event submitted to the safety database in the CRF, or that an event entered in the CRF should be submitted to the safety database. One of the biggest suprises was that a sizeable proportion of CRFs are lacking entries for adverse events that are present in the database, and 35 are missing CRF adverse events log entirely.35
A common issue is failure to report reactogenicity events that exceed the 1-week timeframe, either by hiding them in a covid visit or by simply not making an adverse event log entry.
Fraud narratives
Due to the nature of the CRF submission requirements, the selection received is biased towards bad outcomes. Manipulation is expected and occurred systematically. Here are some of the worst cases:
10011093 - 59 / M / b2 / BMI 28.2 / doses: 29.7 + 19.8
The subject has an adverse event of chest pain, related to “cardiac ischemia”. That’s a bit like coding “lightheadedness” related “limb amputation and subsequent blood loss”. Additionally, there is a covid visit which was created retroactively, as revealed by the CRF on page 401; Pfizer insists on a covid visit on 20.1., and the site replies with “covid illness visit recorded” on 27.1., corresponding to the covid visit being entered on 26.1. on page 322. In the CRF, it does not have an end date, in ADSYMPT the end date is the same as the end date of the adverse event, 15.1. The self-swab dated 21.1. is entered on 26.1.
10131786 - 65 / F / placebo / BMI 34.1 / doses: 21.10 + 12.11 + 25.1
The subject receives dose 3 on 25.1., does not have a dose 4 listed. In the database there are two adverse events, SYNCOPAL EPISODE, 30.1.-1.2., and COVID-19 PNEUMONIA 2.2.-6.2., along with a covid_b illness 30.1.-26.2. with symptoms of diarrhea, cough, shortness of breath and fatigue. There is no covid_a in the database, despite it not being marked erroneous in the CRF after dozens of pages of back and forth. The issue is that covid_b overlapped covid_a (2.2. diarrhea only vs 30.1.-26.2. symptoms see above), the former entered on 19.2., the latter on 3.3. Despite covid_b’s symptoms ending on 26.2. and the visit on the same day therefore being within protocol, there is a deviation logged for “Visit performed outside of protocol specified window.” on 26.2. Notably, the AE audit reveals that the covid pneumonia was “kept in safety due to not meeting efficacy endpoint” and the diagnosis “covid-19” was deleted from the covid illness on 9.3. and that in response to a Pfizer query on 4.3. why the fourth dose had not yet been given, the site reply seems ambiguous as to whether symptoms had in fact concluded by then.
10271191 - 68 / F / placebo / BMI 21.8 / doses: 11.9 + 2.10
The patient is unblinded on 6.1., then has a covid illness beginning on 12.1. resulting in death on 13.2. The CRF file is very disorderly thanks to the site somehow managing to input considerable amounts of data points generated from covid_b into covid_a, which occurred from 17.-23.12. What makes this CRF stand out is that the “Potential ReVax Initial Contact” page recording trial arm assignment and date of unblinding 6.1. was entered on 15.2., two days after the subjects death. Both adverse events were intermittently deleted thanks to a Pfizer query stating the adverse events “met the clinical efficacy endpoint”, only for them to be undeleted ten days after the subjects death. Was this subject really placebo, was he really unblinded and even if he was, would the site have entered a vaccine dose 3 after learning of the subject’s demise?
10471290 - 56 / M / b2 / BMI 36.5 / doses: 24.9 + 13.10
The subject had a positive covid test on 23.2. not counted toward efficacy because of a lack of covid symptoms, despite the subject being hospitalized at the time. This subject has a catastrophic adverse event log involving an abdominal hernia, a laproscopic cholecystectomy (invasive gallbladder stone removal), post-surgical intra abdominal abscess, suspected GI bleed and small bowel strangulation starting on 7.12. with the post-surgical infection having no end date. Covid illness A remarkably has no hospitalisation listed, despite the subject being hospitalized at that time, and there are no symptoms listed despite the abdominal infection starting one day earlier.
10831162 - 30 / F / b2 / BMI 24.7 / doses: 26.8 + 16.9
The subject became pregnant and had a miscarriage. The fraud committed here is the deletion of the adverse events “diarrhea - intermittent” and “nausea - intermittent”. Marked “ongoing? yes” on 29.10., they were deleted on 30.11., when a covid illness was entered with corresponding symptoms, still marked ongoing, until 10.12. when the end date was changed to 1.10. Vomiting is a symptom in the covid visit as well as an adverse event; Pfizer didn’t seem to notice, putting into question why this health outcome apartheid was being done at all. It’s notable the “duplication” of vomiting Pfizer adapted their entire event reporting structure to purportedly avoid didn’t even throw a flag, despite the highly automated system of intermeshing IT structures.
10871150 - 71 / M / b2 / BMI 38.2 / doses: 21.8 + 9.9
An eye-watering 14 adverse event entries don’t tell the whole story of this subject; from 14.9.2020 until 4.3.2021, the only event recorded was “strep throat”, and until they were deleted in favor of covid_a on 21.10., there were also the adverse events of fever and chills. That first covid illness visit in August actually occurred, with data entered a few days later; the second potential covid illness from 1.-5.9 with a visit supposedly occurring on 9.9. yet with no swab is entered into the CRF on 17.11. The only symptom is “other: strep throat”. In March the adverse event log is then expanded with the following: Tonsillar Abscess - Right, Odynophagia, Femoral Hernia - Right, Hypokalemia, Urinary Frequency, Urinary Tract Infection, Worsening of Hypertension, Prominent Reactive Lymph nodes in Cervical Area, Right Anterior Cervical Tenderness, Right Anterior Cervical Swelling, Abdominal Pain. These all have start date 1.9 and end by 5.9. except for the femoral hernia, which has no end date. Cut-and-dry case of lying by omission?
10891182 - 47 / M / b2 / BMI 23.5 / dose: 25.8
This subject only received the first dose, on 25.8., had “worsening of COPD” from 26.-27.8., “jumped from a dumpster” and broke their hip on 9.9., and “worsening of polysubstance drug abuse” and “psychosis” on 27.9. Two adverse events, “viral illness unspecified” and “vomiting” both 27.-28.9. were deleted from the CRF adverse event log on 16.10. after having been entered on 6.10. A corresponding covid visit was entered on 9.10., and included a negative local swab dated 28.9. This covid visit was deleted on 20.10. and the adverse events not reentered into the AE log. The subject’s “lost to follow up” date of 23.11. was entered into the CRF on 25.2. and initially read 12.2., however, there are already CRF comments from 7.10. stating “left blank, potential subject could return to site”. The majority of the subjects’ substantial medical history was only entered in October, there are several queries in the audit with Pfizer asking for clarification and getting next to none.
10951180 - 66 / M / b2 / BMI 28.1 / doses: 31.8 + 22.9
This subject has the adverse events of “post-vaccination myalgia” for five hours on 31.8. and “dizziness”, related to “Atrial Septal Defect”, from 5.-6.11. The medical history reveals that the “atrial septal defect” item start date was initially set to 5.11., one day later it was changed to XX/XX/1953 with the following reasoning: “changed start date to reflect pt's dob since this is due to congenital heart condition at birth”. At the same time, the adverse event term is changed from “dizziness secondary to Atrial Septal Defect” to just dizziness. While the “related” field still mentions the cardiologic origin, this is largely irrelevant as it is not coded. The subject was 66 years old at that point; a congenital heart defect having gone unnoticed so long may happen, but it certainly is peculiar it only became symptomatic after two doses of BNT162b2. The CRF initially has toxicity grade 2 for the heart defect/dizziness, until Pfizer remarks on “Mild '1' per source. Please verify and update, thanks after which the site changes the CRF toxicity grading to 1; the database toxicity grading is 3.
10961355 - 60 / F / placebo / BMI 39.1 / doses: 17.9 + 8.10. + 12.3.
According to the database, this subject received a third dose of vaccine on 12.3. after suffering the events of multiple lower extremity fractures and fall on 31.1. and hypokalemia and urinary tract infection on 5.2., corresponding to AESPID 2, 5, 6, and 7 respectively; as described previously, such gaps in the AESPID listing represent deleted adverse event records. The problem with this subject is that there are no adverse events listed in the CRF, and neither is dose 3. The only entry in the CRF after visit 3 in November is the unblinding on 3.3. There are also no queries from Pfizer requesting the adverse events or the third dose be entered, nor are there any hints whatsoever as to the nature of the deleted AEs.
10971061 - 67 / F / b2 / BMI 22.5 / doses: 28.8 + 17.9
This subject has an “asymptomatic” covid illness while hospitalized for the SAEs of uremic encephalopathy and acute kidney injury 22.-25.10. In database “CE”, subject has “covid-19 like illness” = “no”, and in ADSYMPT all symptoms are marked “no” except for fever, which isnt marked at all, and have neither start nor end date; there is only a negative swab on 12.11., which is also the date of visit 3. The CRF reveals that both “no COVID testing done according to hospital records.” in a comment on 20.11., but also that “Subject had no signs or symptoms of COVID” on 24.11. at 12:08, then querying whether the visit should be erroneous at 15:43. The acute kidney injury is related to “concomitant drug therapy” but there is no closer specification of what drug it was. There are several remarkable items in the adverse event audit: firstly, a Pfizer query claims the adverse events’ initial designation of “altered mental state” began on 28.8.: “SAE RECON: Altered Mental Status(Onset date:28Aug2020) is not reported to Safety database but marked serious on AE CRF. Confirm seriousness and report to Pfizer immediately. If this event is not serious, downgrade the event on AE CRF” on 3.11., and subsequently on “CLINICAL - Reason for SAE status is both Hosp and Medically important; however, SAE report has only Hosp. Please review and harmonize between reports (2 reasons is acceptable).”, revealing that the reported time frame was only for the hospitalisation, but not the symptoms. Remarkably, Pfizer first queries for a covid test on 5.11., to which the site replies “changed information”, and then again on 10.11. to which the site replies “Form will be sent. The patient was not tested for COVID” on 11.11. The SAE report includes: “CLINICAL - SAE report states subject has bilateral apical fibrosis on chest CT; please clarify if a known pre-existing condition and add to Med Hx, or if newly diagnosed, consider as a safety event for potential AE reporting” on 5.11.; on 9.11., the medical history is edited to include ongoing pulmonary fibrosis starting in “Dec/UNK/2015”, despite an entry from 6.11. stating “This was found on CT of the chest performed 22Oct2020. This was unknown prior to that time so it should not be added as history”. Pfizer also queries whether the medical history of depression might be grounds for ineligibility due to inclusion criteria #1, to which the site replies “subject meets qualification .” 25 hours later. The way this case was treated was distinctly abnormal; usually the symptoms are just hidden in the covid visit, not the entire covid visit disacknowledged and the symptoms leading to hospitalisation, apparently ongoing since day of dose 1, simply ignored. This subject seemingly received the second dose in the right arm because of a fentanyl patch on the left deltoid, but Pfizer claimed did not match with records, so the site changed it to the same arm as visit one. There is no mention of why the subject was wearing a fentanyl patch. This error is repeated at visit 3, when the reactogenicity eDiary was evaluated.
11091503 - 64 / F / b2 / BMI 26.9 / dose: 11.9
The subject has an adverse event of abdominal pain right upper quadrant related to “gallbladder disease” with start date Sep/UNK/2020 and end date 30.9.2020, toxicity grade 3 yet not a serious adverse event, but serious enough for the subject to decline the second dose. All medical history items are entered a day after dose one, and only include Asthma, 2012-ongoing, Postmenopausal 2000-ongoing, and Hysterectomy, 2014. In the audit, Pfizer even asks “clinical: pls advise if subject with known hx of gall stones/gallbladder disease and if so update MedHx CRF accordingly. Thanks”, to which the site replies “SUBJECT HAS NO KNOWN HISTORY OF GALL BLADDER DISEASE”. While the database lists ADVERSE EVENT discontinuation on 30.9., the “withdrawal of consent” form in the CRF was last changed to “not applicable” on 7.1., and there is even discussion about the Revax contact on 4.1., entered on 11.2. with queries and responses til 3.3. What happened in between CRF data cutoff and database construction that made Pfizer cut the subject’s data so early?
11141006 - 73 / M / b2 / BMI 27.9 / doses: 12.8 + 1.9
The subject has the adverse events of ventricular tachycardia, blood coagulation disorder, hypokalemia and pulmonary embolism. The medical history shows not only removal of an eye cyst on the day of dose 1, there is also an edit on October adding “recurrent angina” ongoing since 2005, and then another edit in March adding ejection fraction below 30% and “AUTOMATIC IMPLANTABLE CARDIOVERTER DEFIBRILLATORS IMPLANT”, both in 2011. The adverse event of “ventricular tachycardia” has toxicity grade 4, required surgical intervention, lasted four days to 15.12. and involved defibrillation by the implant. Hypokalemia begain 12.12. and lasted until 20.12. The pulmonary embolism occured on 7.1. and was resolved on 10.1. It was entered on 13.1. as "related to “blood coagulation disorder”. Five days later, Pfizer queried whether this is a new diagnosis or whether it should be added to medical history. Two days after that, the site initially submitted an SAE followup external to the CRF, to which Pfizer replied “clin: requested info not seen in Argus. Pls provide start date of coag disorder. If prior to enrollment should be added to MedHx CRF, if after pls report as separate AE. Thanks“. It then takes another week until the site finally entered the coagulation disorder into the CRF on 28.1. In the audit of that adverse event, Pfizer initially queried “clinical: pls confirm this is a new diagnosis and advise if there is a more specific diagnosis that can be made. Is platelet count normal? Thanks“. The site replied “Original value is correct”. Pfizer queried “clinical: answer is non responsive, pls confirm this is a new diagnosis and advise if a more specific diagnosis is available eg was subject hypercoaguable? and if so, how was the dx made? Was the platelet count normal? Thanks” to which the site ultimately replied “This is a new diagnosis and there is not a more specific diagnosis platelet count is unknown to site” on 3.2.
11161059 - 20 / F / b2 / BMI 40.1 / doses: 31.8 + 22.9
The subject had adverse events of pancreatitis with gallstones and bile duct stones between dose 1 and 2, staying inside the dosing window of 19-23 days despite the obstructive pancreatitis event ending three days before dose 2 and needing two surgical interventions according to the AE audit. The database also lists unprotected sex with HIV-positive male and a pregnancy. The CRF lists a deleted adverse event of sinus arythmia, related to “This was likely there before and just undiagnosed”, entered on 1.10. with start date 14.9., confirmed as ongoing on 26.10., and deleted on 24.2. due to “changed information”. There is also a mysterious Pfizer query regarding an adverse event of hepatitis: “SAE RECON:AER#2020363544Hepatitis (onset date:14Sep2020)was reported as serious in Safety database but missing in AE CRF. Please confirm and update CRF. If safety update is required, submit a follow-up SAE Form.” followed by the same query and adverse event number but “pancreatitis”, followed by “Event terms updated in SDB, Hepatitis no longer listed. New query issued for new event issues.”
11171167 - 82 / M / b2 / BMI 28.1 / doses: 25.9 + 14.10
The subject had an adverse event of vertigo from 6.-9.11., toxicity grade 3, related to “ETIOLOGY UNKNOWN”. The adverse event audit log reveals the adverse event was entered over a month after it occurred (14.12.), that an ECG showed bradycardia, and that Pfizer was querying aggressively for a covid visit. Notably, the subject’s medical history was updated with “elevated prostate specific antigen” ongoing since 2014 on 17.11.
11181031 - 78 / F / b2 / BMI 17.9 / doses: 14.8 + 4.9
The subject had an event of “chest pain” in the CRF, changed to “cardiac chest pain” in the database. The audit reveals the event involved cardiac catheterization and a failed stent placement, and was entered into the CRF in February after occurring 24.-26.11.
11281103 - 48 / F / b2 / BMI 22 / doses: 12.8 +1.9
The subject had adverse events of kidney stones, severe hypokalemia and e.coli urinary tract infection from 3.-22.10. and a potential covid illness with the same start date but ending on 17.11., including all possible symptoms except fever, cough, and loss of taste and smell. A Pfizer query on 1.11. states: “Clin: My apologies for the mistake, since GASTROENTERITIS is a SAE with a negative SARS-CoV-2 result should be documented on the COVID-19 CRF ILLNESS DETAILS form and should be also captured on the AE CRF page, please do not delete from the AE CRF form.” to which the site responds the next day with “per DM, SAE has been undeleted“; the AE audit shows that on 13.11., Pfizer queries: “SAE RECON: AER#2020405778,the term was updated to 'Escherichia coli urinary tract infection' in Safety database while recorded as 'GASTROENTERITIS' in AE CRF. Please confirm correct term.If safety update is required, please submit a follow-up form”, to which the site responds by completing the change seven hours later. Neither do the dates change to record the actual symptom duration, nor are further adverse events added to cover the scope of illness. The covid illness is also abused by extension of symptom duration instead of additional visits being completed; the symptom stop date was initially “ONGOING” when entered on 30.10., then changed to “17.10.” on 6.11., back to “ONGOING” on 17.11., then changed to “22.10.” on 23.2. at 12:23 and to its final value of “17.11.” 6 minutes later at 12:29.
11291260 - 17 / F / placebo / BMI 22.1 / doses: 20.11 + 15.12 + 25.1
The subject had a life threatening anaphylactic reaction to dose 3. There were initially two adverse events, hives left arm and shortness of breath, that were consolidated on 12.3. The allergic reaction seems to have begun about 36 hours after receipt of vaccine. The audit contains the remarkable statement “per Clinical Trial Manager Lindsay Kevles, "Per the safety team, the symptom “Shortness of breath” experienced by the subject was due to an allergic reaction to the vaccine. This is specific situation, we do not expect a nasal swab to be collected.”, serving as a reminder just how closely Pfizer was micromanaging the trial.
11401020 - 51 / F / placebo / BMI 34.4 / doses: 3.8 + 25.8 + 15.1 + 5.2
The subject had an adverse event of “benign left groin tumor” with start date 7.12. initially entered on 15.1., the day of dose 3, which is changed to “B-cell lymphoma” on 10.2., five days after her fourth dose. The timeline is unmistakeable in that the malignancy developed after vaccination, yet this distinction is not made, resulting in the database showing Pfizer give someone with a b-cell lymphoma two vaccines.
12311182 - 63 / M / b2 / BMI 31.2 / doses: 14.8 + 4.9
this subject only has an AE of ischaemic stroke from 7.12.-10.1., yet the audit reveals there is additionally an event of pericarditis mentioned several times “Clinical SAE report on 16Dec20 has the discharge date of 09Dec20 in the narrative; however, Hospitalization details for event of Pericarditis has admission/discharge on 09Dec20”. This event was not deleted from the CRF as it wasn’t even entered in the first place.
12312390 - 25 / F / b2 / BMI 38.1 / doses: 19.8 + 9.9
The subject had an adverse event of “osteochondritis”, yet the audit reveals this should actually be “costochondritis” - chest wall pain. For some undiscernable reason, it stays the way it is: Pfizer query 14.9.: “Chest wall pain may be costochondritis, rather than joint pain c/w osteochondritis. Please review safety event term and update if required.” Site reply 18.9.: “The term was reviewed by the team SAE report manager and the assigned term will be maintained. Thank you.” It’s also worth mentioning the subject’s covid illness was initially from 10.-18.9. (beginning one day after dose 2) with a visit mysteriously occurring on the 16.9., yet no central lab swab, when it was entered over a month late on 30.10. This start date was changed to 16.9. on 18.2. The covid convalescent visit was entered on 13.10., a week before the covid visit entered the record.
12312982 - 36 / M / b2 / BMI 25.2 / doses: 21.8 + 9.9
Augusto Roux’ subject ID, who was recently called upon to be Argentina’s state prosecutor in a criminal case against Pfizer. Much has been written about his case and the medically confirmed vaccine-induced pericarditis fraudulently recorded as anxiety under the responsibility of Fernando Polack, lead author of the NEJM publication. The CRF is rife with oddities. The covid illness from 9.-12.9. is fake, retroactively entered on 27.10, and includes the very much covid-unrelated symptoms of dark urine and dizziness. They had the bright idea of using the dose 2 swab for the covid visit but ultimately refrained from doing so. Despite the purported consent withdrawal on 23.9. entered on 6.10., the adverse event of “anxiety” has an end date of 14.10. A query from 18.2. also states that: “I clarify that although the DISPOSITION - FOLLOW-UP date is SEP 23, 2020, at the request of the sponsor the information on the end date was compiled by the PI”, in contrast to an entry on 17.11.: “according to medical record, the 14/oct/20 was the date that the volunteer went to the hospital to sign the receipt of the certified letter that details his branch of treatment, retiring in excellent spirits, constituting your last contact, thank you”. At the time it was initially entered on 10.11., the consent withdrawal date was 29.10. In the audit of dose 2, there is the astounding quote “SAE was a febrile syndrome secondary to severe reactogenicity related to the investigational product. Thank you.“ from 22.9. - yet the adverse event log lists “suspected covid illness”. The adverse event audit even repeats this: “Taking into account the data of the epicrisis, having ruled out Sars-Cov2 disease, the clinical picture of fever, myalgia, headache and general malaise is interpreted as a febrile syndrome secondary to severe reactogenicity related to the product under in[vestigation]“ on 21.10. The adverse event term changed from bilateral pneumonia on 14.9. to febrile syndrome on 22.9. to suspected Covid illness on 11.10., to “Covid illness” and back to “suspected” on the 12.10. On 3.10., a “Mabel Berrueta” enters: “Based on medical Record, Dr Polack PI of the study wrote on 17SET2020 that "Hospitalization is an SAE not related to the vaccine, due to suspicion of COVID-19". Cause is unknown.” On 22.9., the SAE causality was changed to “RELATED” after Pfizer asks why it was reported as related in the SAE form but unrelated in the CRF, and back to unrelated one day later. A “subject safety-related” unblinding on 6.10. was entered on 9.10. and deleted on 1.3.
12313184 - 48 / M / b2 / BMI 24.6 / doses: 22.8 + 10.9
The subject has an adverse event of right carpal tunnel syndrome from 15.10. to 19.2. listed in the database. The audit astonishingly enough reveals it was infact bilateral, with surgeries in February and March. A 7.2. site response to a Pfizer query states: ”Elective surgery was scheduled in the right hand and a second time left hand. performed on FEB 02, 2021. The pacient has got history of cholecystectomy due to gallstones.” The only medical history item of cholecystectomy 1.1.2016 was added three days earlier with no apparent prompt on 4.2. - was this listed to avoid entering an adverse event?
12314035 - 63 / M / b2 / BMI 28.7 / doses: 25.8 + 14.9
The subject had adverse events of “anxiety crysis” (6.5 hours on 29.9.) and “angina pectoris” (20.-24.9.) listed. The CRF reveals these events were actually acute myocardial infarction (renamed to Angor and then Angina on 9.10., and finally to Angina pectoris on 13.10.) and dyspnoea, the latter being changed to anxiety after Pfizer query bullying for a covid visit:
13.10. “The patient presented dyspnea, which was considered an adverse event since he had presented as an Acute Myocardial Infarction and he consulted for that reason”
22.10. 9:41 “Clarification: "Dyspnoea" was a typing error. Form updated”;
22.10. 14:22 “CLINICAL On review of the SAE report for angina, an event of 'SOB functional class III' has been mentioned, yet it does not appear on the CRF. Please review for safety and update AE log CRF as appropriate.”
12314335 - 65 / M / b2 / BMI 35.2 / doses: 26.8 + 14.9
The database lists this subject as having “deafness neurosensory” with empty AETERM in ADAE, with a description as “Bilateral moderate sensorineural hearing loss” in the narrative, lasting from 20.9. to 22.2. There are no adverse events logged in the CRF, and there is not a single non-automated query in the entire audit.
12321299 - 55 / M / b2 / BMI 23.7 / dose: 6.10
The subject has an adverse event of hypertension in the database with start date day of dose 1, no second dose due to treatment discontinuation on that same day, and study discontinuation on 29.10. as “lost to follow up”. The CRF adverse event log was never started, and “undiagnosed hypertension” with start date 6.10. was added to medical history on 12.3. with the explanation: “Illness was first noticed at V1. Patient was instructed to see PCP.” An exchange from the CRF’s “end of treatment” form on 10.3.2021:
04:53 AM: “CLINQUERY: Please clarify reason for lack of Vax 2 - per comment 'investigator decision'. Was any event linked to this decision?”
10:32 AM: “Pt blood pressure was too high to receive vaccination”
13:39 AM: “CLINQUERY: For high BP - there is no adverse event entry nor prior med hx for this occurrence. Please review if one of the listings needs updating. Per your information was a temporary delay initially intended?”
11.3.: “Please unlock Med Hx. Temporary delay was initially intended.”
“Subject did not receive V2 vaccination due to investigator decision and was then lost to follow up” The date in the CRF remains day of dose 6.10., Pfizer changed the date to 29.10. LTFU is entered on 27.1., along with the dose 2 visit as “not done”.
12411471 - 34 / F / b2 / BMI 23.6 / doses: 25.8 + 15.9
The subject has a diligently recorded list of reactogenic adverse events including lymphadenopathy and six days of injection site swelling after dose two, and an adverse event of “left mumps” with start date “Feb/UNK/2021”, related to “sjogren syndrome”. The site’s answer to a Pfizer query for clarification on 11.3.: “This was suspected and is not related to a pre-existing condition”. As noted in a previous case, the “related to” field is only window dressing and is not coded or taken into account.
12511060 - 33 / M / b2 / BMI 51.2 / doses: 26.8 + 16.9
The CRF claims the subject is placebo, in direct contradiction to the database. This discrepancy is not resolved despite signatures dated 12.3. According to the database, the subject had an adverse event of nasal congestion from 2.-4.9. and “exposure during pregnancy” of his wife with start date Jan/UNK/2020. The pregnancy is not in the CRF despite a ReVax contact on 9.2. entered 23.2. There are two deleted adverse events of chills and sore throat both lasting from 6.-8.10., created and deleted on 22.10.; there is a covid illness with cough as the only symptom from 9.-11.10. entered on 21.10. There is a second covid illness entered one day after end of symptoms, yet the swab is one week out of window for a long list of reasons including subject error and error by the swab logistics company Marken. The adverse event of nasal congestion is curiously marked related, and only entered after a Pfizer covid visit query on 23.10.: “Clinical Query: Per reported PD, nasal congestion reported via phone on 02Sep was not entered into InForm. If this was a symptom of potential COVID, please record in COVID Illness Visit. If not, please record on AE CRF.”
12511239 - 48 / F / b2 / BMI 43.8 / doses: 13.10 + 2.11
The CRF lists the subject as placebo, the database lists as BNT. The database lists an adverse event of myocardial ischemia 13.-16.11. The CRF additionally has a deleted adverse event of chest pain, related to “Previous Chest Pain Secondary to Congestive Heart Failure” starting Aug/UNK/2020 and an end date of 30.10. first entered on 3.11. and deleted on 18.12; one day earlier on 17.12., the medical history was edited to include chest pain ongoing since Aug/UNK/2020. There is a covid illness entered on 18.11. along with the heart attack AE and same dates with an “indeterminate” local swab from 13.11. (“Results are unknown.“) and a central lab swab on 20.11.; however, the barcode entered into the CRF does not appear in the database, and there is no swab associated with the visit in ADSYMPT. Other subjects have swabs much later relative to symptom end than that in the database, leaving the impression this might be a suppressed covid-positive case.
12511262 - 32 / M / b2 / BMI 33.5 / doses: 27.10 + 17.11
The database lists the adverse events seizure on 15.12. related to “methamphetamine usage 12/13/20 hx of afib” and moderate dizziness from 13:48 to 14:50 on 18.12., related to “possible vasovagal response”. The following medical history additions occur: gout ongoing since 1.8.2020 is added on 19.11., and hypertension ongoing since UNK/2007, epilepsy ongoing since 1.4.2018, and atrial fibrillation ongoing since UNK/UNK/2020 are added on 28.12. The a-fib item is edited to have a start date of 15.12.2020 ten minutes after being entered, then changed to “hx [history] of atrial fibrillation” one day later, and then to its final start date UNK/UNK/2018 on 31.12.: “Subject informed site via telephone that onset began in 2018.”
12521010 - 80 / M / b2 / BMI 29.2 / doses: 17.8 + 8.9
This subject died within one day of a covid diagnosis with a positive protocol-compliant Abbott local test, yet is not counted towards efficacy. Data from the covid illness reveals an enlarged heart and oxygen saturation of 89% but a lack of covid-typical lung consolidations. There are two sets of lab work, with the deleted set having tighter ranges and correspondingly worse readouts. There is also an adverse event of scalp laceration related to “accident” in the database; the CRF reveals the subject fell over in a chair, so there should be an adverse event of fall coded additionally.
12601108 - 42 / F / placebo / BMI 29.8 /doses: 10.9 + 1.10 + 14.1 + 1.2
The CRF only lists AESPID 1 and 5 with 2,3,4 deleted, while the ADAE entries include AESPID 6, 8, 9, 10, and 11; AESPID 7 was added and deleted after CRF data cut. AESPID 5-11 all share the same start date of 9.3., making it all the more absurd only the ankle fracture is included in the CRF. The AEs not reported in the CRF and their corresponging AESPIDs are fall (6, related to “subject fell and injured ankle”, 9.3.>ongoing), acute encephalopathy (8, related to “complicated UTI, metabolic hypothyroid and possible side effects of gabapentin”, 9.-10.3.), Urinary Tract Infection (9, related, 9.3.-12.3.), hypotension (10, related to “received fluids as maintenance until discharge to correct hypotension”, 10.3.>ongoing) and Altered Mental Status (11, related to “UTI, acute encephalopathy”, 9.-10.3.). There is no indication why the subject might be taking gabapentine; the only medical history items are thyroidectomy in 2000 and Graves’ disease ongoing since then, both entered along with all the other visit 1 data on 12.9. The deleted adverse events of muscle pain, runny nose and dry cough all from 5.-7.10. were created on the day of visit 3, 29.10, and were initially all assessed as related due to being within 7 days of dose 2. They were deleted one day after this query on 12.11: “CLINQUERY: Please confirm if this AE is the same as a COVID illness symptom reported on SOD (new/inc cough). If yes, please consider if AE should be removed from AE CRF per study team directive that COVID symptoms should be captured on SOD form only”. The corresponsing covid illness visit was entered on 15.10. with a central swab and a negative local swab from UMass Memorial Medical Center on the same date, the latter of which the site was “advised to delete” on 22.10. Notably, the unblinding date of 8.1. entered on 12.1. is changed to 16.12. on 3.2. following a query on 1.2.: “Per source the initial contact was made 16DEC2020 via email. Confirm this date.”
44441422 - 65 / M / b2 / BMI 32.3 / doses: 22.9 + 12.10
This subject had a positive swab at baseline. According to the database, the subject has an adverse event of “pain in left heel” for six hours on 29.8. and “Poorly differentiated colon adenocarcinoma” on 20.1. with no end date. There is a covid illness from 15.-16.12. in ADSYMPT with a diagnosis of “acute diverticulitis” with toxicity grade 1 in CE. The CRF reveals the “covid illness” had a start date of 3.12. for two weeks and was initially marked ongoing on 17.12., with symptoms of fever and “abdominal pain”. The diagnosis of acute diverticulitis was entered on 13.1. The adverse event of cancer is not reported in the CRF despite ReVax contact on 2.2., and there are no reminders or queries related to it, so the CRF adverse event log marvelously contains only the heel pain (related to a bone spur, imagine havíng that all your life and it hurts only just after being jabbed - weird) with no mention of cancer or diverticulitis.
44442319 - 41 / M / b2 / BMI 25 / dose: 27.9
Subject discontinued treatment and study due to “worsening of panic attacks” on 2.11., entered on 11.11.: “Due to a psychiatric condition (panic attacks), subject expresses his desire to withdraw consent”. The discontinuation was indeed marked as “consent withdrawal” instead of “adverse event” until 2.12., one day after the solitary medical history entry of panic attacks, ongoing since 2015, was created, as announced on 27.11.: “This AE was submitted extemporaneously as the query requested. The subject has already withdrew his consent, so the AE cannot be followed-up. Also, his baseline psychiatric condition will be submitted shortly as a Medical History report. Thank you.” Reminiscent of the way the site also handled Augusto Roux’ case.
Injury narratives
During my reading i tagged a total of 63 subjects with “rekt” for having adverse event logs indicative of not having had a very nice time, to put it lightly. This is not a tag I used consistently enough to really analyze, but the individual suffering should be recounted. Average age 62.06 years, 39 male, of whom 24 are BNT subjects and 15 placebo subjects, 6 with no unblinding doses and 24 female, 12 BNT, 12 placebo, and only one female placebo subject with no unblinding doses.
11141080 - 61 / M / b2 / BMI 37.1 / doses: 24.8 + 30.9
This subject weighed in at 127 kg despite having no legs; the medical history lists right and left leg traumas starting 1.1.2005 and ending 1.11.2012 and 1.1.2018 with respective amputations. There is also type 2 diabetes and neuropathy since 1986, along with a laundry list of medication intensive conditions like hypertension, anxiety, hyperlipidemia and “[Recurrent Methicillin-resistant Staphylococcus aureus Infections]”, ongoing since January 2017. 5/12 medical history items including the recurrent stump infections were entered into the CRF on 13.10. The delay between doses is due to hospitalisation for the following list of adverse events: MRSA infection Right Stump, Atrial fibrillation, chest pain, Pulmonary Hypertension, Left Ventricular Hypertrophy, mitral valve regurgitation, Tricuspid regurgitation, acute kidney injury, ‘skin avulsion, left finger’, bilateral hand pain. All events start on the 12.-14.9, the hand pain and finger injury end 1 respectively 2 days later, acute kidney injury ends 2.11., and the stump infection ends on 8.10. The other bolded events have no end date.
11111010 - 75 / M / placebo / BMI 29.3 / doses: 31.7 + 21.8 + 5.1 + 26.1
This subject’s database adverse event log reads as follows: clostridium difficile infection (13.3 > ongoing), Nonsmall cell lung carcinoma stage 3 (25.2 > ongoing) Acute hypoxic respiratory failure, Acute kidney injury on chronic kidney disease stage III, Urosepsis (all from 11.-15.1.). This subject had a positive covid_a illness with chills and fever from 4.-5.10. The audit reveals that the lung masses were benign from 2019 until six days after the first dose of vaccine in an entry from 11.3 : “No, the masses were added to medical history with start date of 2019. At that time, masses were benign. Cancer is new diagnosis.” “Formal cancer diagnosis was on 11Jan2021 per medical records. Does not make sense to change the date of the masses that patient had for years prior at least since 2019. This date will not be changed.” The adverse event that made it into the database has a start date of 25.2 because the initial AE was deleted for not being considered serious (AESPID 4 and 5); “clinical: pls confirm assessed to not meet seriousness criteria. Thanks” “Assessment done and this AE is not considered serious” - successive queries from the deleted AESPID 4, entered on 26.1. The subject also has a lymphocyte count of 0.31 with a range of 1-5 in a set of hospital labs from 11.1., visible thanks to a covid_b illness created at Pfizer’s behest: the swab is collected 6 days after end of symptoms and one day after the covid illness is entered into the CRF; “Safety team requested the swab collection”.
The concept of vaccine-induced antibody-dependent enhancement has been discussed widely, but vaccinating someone who’s already been infected is not only silly from an immunological perspective, due to non-S epitope exposure providing vastly superior immune responses than monodominant spike, could be an additional risk factor for severe adverse events like this subject experienced.
12141018 - 30 / M / b2 / BMI 25.5 / doses: 2.11 + 23.11
This subject suffered a recurrent vertebral disc hernia on 18.11 lasting til 2.12 requiring surgical intervention. The only medical history item of previous hernia surgery is entered into the CRF on 23.11. The subject then suffers a pulmonary embolism three days after surgery on 5.12 and gets put on twenty days of twice-daily dose escalating Clexane injections and Xarelto, both aggressive blood thinning therapies. There are deleted adverse events of “spinal fusion surgery” and “Worsening of vertebral disc herniation”, both on 2.12. Along with the embolism being related to “immobility and surgery”, the massive health impact suffered by the subject becomes clearer.
10661242 - 54 / M / placebo/ BMI 33.4 / doses: 11.9 + 2.10 + 18.1 + 10.2
This subject had a positive covid illness with symptoms ending ten days before receipt of dose 3, and developed bilateral pulmonary embolisms five days after dose 4. The toxicity grade was changed from 4 to 3 after a remarkable Pfizer query: “Clinical - Severity grade is 4; however, SAE is no longer Life Threatening [per protocol Sect 10.3.3 Assessment of Intensity, Grade 4 = Life threatening]. Please update toxicity/severity grade”
There are many more narratives unquestionably worthy of being included here, perhaps even more so than the ones chosen, but I made the decision to prioritize description of fraudulent practices.
Trial arm ambiguity
Arguably the most important data point in assessing a clinical study’s outcome is beset by critical errors in the case of C4591001. While there is a subset of placebo patients with adverse event profiles that are difficult to accredit to i.m. administration of saline, these doubts only have weight thanks to the presence of at least eight subjects with discrepant trial arm assignments between CRF and database - the database says one thing and the CRF says another. As with other anomalies, this list represents a lower bound.
A software-based analysis of trial arm assignment is in progress, yet not complete in time for this article. The following subjects have or had unmatching trial arm assignments in the CRF and the database:36
Note: “instead of” and “corrected” in this context refer to the database value; we have no way of knowing which is indeed correct
10131084 (full error, CRF states placebo instead of BNT)
10191215 (full error, CRF states placebo instead of BNT)
10841480 (full error, initially “not willing” changed to BNT 44 days after entry)
10871286 (full error, CRF states placebo instead of BNT)
10931128 (full error, CRF states placebo instead of BNT, even received dose 3 but was discontinued after dose 1 so 2 doses total)
12511029 (full error, CRF states placebo instead of BNT)
12511060 (full error, CRF states placebo instead of BNT)
12511239 (full error, CRF states placebo instead of BNT)
10131229 (wrong final value despite “changed” according to site, CRF states BNT instead of placebo)
10721037 (was correct initially, switched to BNT 4 days after entry and corrected to placebo 30 minutes later)
10721064 (was correct initially, switched to BNT 4 days after entry and corrected to placebo 1 hour later)
10131517 (corrected to BNT 12 days after entry)
10391038 (corrected to BNT 6 days after entry)
10461046 (corrected to BNT 6 days after entry)
10471194 (corrected to placebo 7 days after entry)
10561022 (corrected to BNT 30 minutes after entry)
10791183 (corrected to placebo 12 days after entry)
10801035 (corrected to BNT two days after entry)
10801059 (corrected to BNT six days after entry)
10821083 (corrected to placebo 23 days after entry)
10951173 (corrected to BNT 6 days after entry)
11141075 (corrected to placebo one hour 15 minutes after entry)
11171141 (corrected to placebo <30 seconds after entry)
11311161 (corrected to placebo 6 days after entry)
11401066 (corrected to placebo 29 days after entry)
11421044 (corrected to BNT 46 days after entry)
11421073 (corrected to BNT 37 days after entry)
11421202 (corrected to BNT 32 days after entry)
11421215 (corrected to BNT 17 days after entry)
11781025 (corrected to BNT 1 minute after entry)
11971097 (corrected to placebo 15 days after entry)
12101029 (corrected to placebo <20 seconds after entry)
12121024 (corrected to placebo 4,5 hours after entry)
12171044 (corrected to placebo after 3 days)
44441771 (corrected to placebo <20 seconds after entry)
The following subjects have adverse events in close temporal proximity to administration of placebo:37
10031111 - developed an adverse event of related psoriatric arthritis from placebo
10071315 - dose 2 delayed due to corticosteroid treatment of hearing loss developed after dose 1
10151134 - had months of vertigo after dose 1 placebo, only received one dose of placebo
10151255 - had four days of vertigo after receipt of placebo
10221024 - developed a related lymphadenopathy for almost two months
10271055 - developed unilateral limb weakness the site attributed to gastroesophageal reflux disease
10471252 - developed 100% occluded right coronary artery four days after dose 1
10541067 - developed a serious anaphylactoid reaction to placebo
10681066 - developed lymphadenopathy from dose 1 placebo
10871228 - adverse event of acute heart failure five days after dose 1 placebo
10871354 - September adverse events of fatigue, myalgia and soreness in vaccine arm with no end date entered March
10901415 - discontinued after dose 1 due to adverse event of headache which was deleted and moved into a covid visit
10901507 - discontinued due to allergic reaction to placebo dose 1
11121122 - developed diarrhea and other symptoms in response to both doses of placebo
11121337 - discontinued study due to fatigue, diarrhea, headache, myalgia, nausea and vomiting for a week after dose 1
11201127 - one month of diarrhea after second dose of placebo
11201432 - had redness and swelling at injection site and other reactogenicity symptoms, later developed pancreatic cancer
11281267 - five adverse events related to dose 1, including muscle pain with toxicity grade 3
11311095 - developed lymphadenopathy from dose 2 placebo
11331537 - bilateral lymphadenopathy after placebo dose 2
11401078 - diarrhea after both doses of placebo
11451059 - headache, photophobia and injection site pain in response to placebo
11521551 - 101.9 °F fever after dose 1 placebo
11631059 - allergic reaction to placebo
11661047 - allergic reaction to placebo
12171044 - lymphopenia one week after dose 1
12241065 - discontinued after Fatigue, Feverish Chills, Irregular Heart Rate, Diarrhea, Increased Blood Pressure from dose 1 on 24.8. til 2.-10.9.
12261067 - adverse event of pain at injection site for one day on 2.9. yet reactogenicity in CE shows injection site pain from 1.-5.9., along with other symptoms
12261094 - several days of diarrhea after both placebo doses along with other symptoms
12311946 - one week of related headache + toxicity grade 3 syncope after dose 2
12312868 - suffered a fall one hour after receipt of placebo
12471121 - severe fever and fatigue after each dose of placebo
12601035 - 27 days of headache from dose 1 placebo
44441761 - developed a rash after receipt of placebo
There are also placebo patients with an unblinding date but no dose 3 date and no related queries.38
“Decoy” CRFs
There are at least 6 subjects39 whose CRFs were erroneously included; somehow “safety narrative” flags were attached to these subjects by mistake, and this mistake was just followed through on all the way to BLA submission. It has to be assumed this was corrected at some point because there is no evidence that it was.
10371141, a 61 year old black woman with a medical history of hysterectomy was found to not, in fact, be pregnant,40 10901492 didn’t discontinue due to one day of fatigue41, neither did 10911297 (despite it being 4 days of fatigue),42 10921021 didn’t discontinue because of two days of body aches43 , one day of chills turned out not to be enough to dissuade 12541006 from further participation,44 and 12541189 was undeterred by one day of headache.45 While all adverse event discontinuations have a CRF file, only 4/28 subjects with “physician decision” discontinuations do.46
Except for the first mistaken pregnancy, these were all supposedly safety-related subject withdrawals. As previously mentioned, there is one auspicious pregnancy in the database which should have a CRF but doesn’t; following that train of thought, are there 5 safety-related subject withdrawals which should have a CRF, but don’t?
JPG CRF files
By file size, 11 subjects47 stand out from the rest due to their filesize; this is du to their contents being all images instead of searchable text. There are additionally 68 CRFs48 where single or multiple pages are non-searchable images. Before OpenVAET’s investment of dozens of hours of his time, I had only identified five of these, so make sure to check out his article49 on the JPG CRF files, too!
These manipulations are a known ingredient of Pfizer-BioNTech’s toolbox. A prominent example is the bnt162-01-interim3-reports.pdf file, which contains 5 study reports detailing the intracellular cytokine staining and SARS-2 peptide stimulation data generated in the BioNTech study. The first version, included in the first submission, weighs in at 73 megabytes compared to the second, Submission 7 version with 25 mb, due to dozens of pages of raw data being in non-copy-pastable format, forcing the regulator to demand a machine-readable version.50
I keep saying CRF but the actual designation is eCRF, a purely digital case report form as opposed to the fully “analog” paper CRFs in use up until the end of the 90s. This makes the intentionally adulterated files all the more suspect and worthy of explanation.
There is a concerning pattern in the distribution of JPG pages; all CRFs of sites 1001 - 1007 have 1-19 JPG pages except for subject 10071101, the only subject from these sites to die.
To make matters more complex, there are JPG pages and.. JPG pages. OpenVAET’s tool generates an HTML repository of links to the individual pages. The first four sites’ CRF pages are all intralinked - the blue underscored links to different parts of the CRF are functional in comparison to most other CRFs, so a click takes you directly to the audit or to the form comments, despite the page not being machine-readable. It also seems very schematic: of 173 JPG pages across 38 subjects for sites 1001, 1003, 1005, and 1007, only 21 pages across 7 subjects contain any data. The 152 pages without data break down like this:
Transfusions: 37 pages
Radiation treatment: 26 pages
Vital signs - Covid: 16 pages
Vital signs - Pulse Ox Room Air : 16 pages
Illness Details - Severe: 13 pages
Hospitalization details: 1 page
Imaging: 14 pages
Medication error: 2 pages
Microbiology specimen: 2 pages
Oxygenation parameters: 15 pages
Respiratory treatment: 11 pages
The 1001-1007 pages with data break down as follows:
Adverse event report: 1 page, subject 10031111. This is the non-audit adverse event report for an event of “related” psoriatric arthtritis in a placebo subject.
Hospitalization details: 5 pages 10031113 10031113 10051293 10061176 10061176
Vital signs - covid: 5 pages 10031113 10051293 10061176 10071306 10071306
Vital signs - Pulse Ox Room Air: 5 pages 10031113 10051293 10061176 10071306 10071306
Oxygenation parameters: 2 pages 10031113 10051293 (Arterial blood gas measurements strongly suggest intubation).
Illness Details - Severe: 1 page 10051293
Imaging: 1 page 10051387 An x-ray with normal results.
Transfusions: 1 page 10071192
Working hypothesis: these pages were inserted or otherwised changed post-facto due to changes in CRF design. Sites 1001, 1002 (no CRF subjects), 1003 and 1007 were responsible for Phase 1, Site 1005 being the odd one out as it only started enrolling subjects in August. 10071101 is the only CRF without any JPG page replacements because the CRF file was created when these changes were already in effect for all files - a death hidden until it was time for BLA submission? This doesn’t explain why some subjects only have one of “radiation treatment” or “transfusions”, despite no data in whichever is missing. It’s also entirely intransparent why these CRFs are intralinked and the others aren’t. An important point is that none of these JPGs are on audit pages, which is where the dirt is. Hypothesis over.
The JPG subjects not belonging to sites 1001-1007 are less ambiguous. Excluding the 11 fully-image CRFs leaves 19 CRFs with 1-22 image pages:
10151089 - page 3 with subject ID, date of birth, ethnicity and gender. Significance unclear
10161042 - the very last page showing when the “casebook signature” was first done, in this case showing 10.9, 14 days post dose 2. This is problematic because it means the PI first signed off on the subject five weeks after enrolment.
10271191 - several pages of the subject status form detailing the separation of “acute hypoxic respiratory failure due to covid-19” into two separate causes of death. Significance unclear; Pfizer closes and reopens the query three times before the site reacts.
10301110 - five pages in total related to a positive covid illness and separate subsequent strep throat SAE. Incredible amounts of back and forth, including rather harebrained changes to the covid illness diagnosis over the course of more than a month.
10441152 - the single JPG page is a correction in the medical history section; “Left Breast Cancer Treatment — Radiation” is changed to “Left Breast Cancer” one day after entry on the 24.10. This subject was HIV positive, yet was enrolled without current labs, and the labs drawn after enrollment led to discontinuation. A new onset breast cancer dated 28.12. was entered 8.3.
10471012 - the JPG page is the 11.3. signature page with a redacted PI name.
10891182 - the subject was lost to follow-up on 23.11. according to the database, yet the audit shows the form initially had 2.10. as LTFU date, entered on 6.10. The first JPG page details the deletion of the data along with the site entry “left blank, potential subject could return to site”. The other two JPG pages are the audit of adverse event “vomiting” which was deleted in favor of a potential covid illness.
10901140 - this subject’s visit 1 data was entered one day after the fact and the blood draw (initially recorded as “not done”) and swab barcodes entered another day later. The medical history entry of type 2 diabetes ongoing since June 2020, entered along with all other data from visit 1, is also in image format. The data entry takes about 35 minutes in total.
10931128 - the visit 1 “date of visit” audit trail page containing discussion between Pfizer and the site regarding adverse event terminology. Even though atrial fibrillation and cardiologist checkups are referenced, the event is ultimately coded as “worsening hypertension”.
11101367 - the audit page listing barcodes of a February visit in the context of asymptomatic surveillance swabbing.
11161045 - the “vital signs - covid” form comment of “N/A” entered on 14. and 26.10. Seeing as there is a concurrent UNPL (unplanned) visit recorded where vital signs are measured, this is omission of potentially damaging data. This placebo subject had terrible health outcomes as detailed more closely in the narratives section.
11261244 - this subject has a whole 22 JPG pages, and also stands out for not having any adverse events recorded in the CRF, yet the database contains four adverse events after unblinding, “Chills” “Fever” “Left upper arm injection site pain” “Lymph node swelling left armpit”, all related and occurring after dose 3. The first block of image pages begins in the reactogenicity eDiary evaluation audit for visit 3, covers the concomitant medications - vaccination page (Menveo MenACWY meningococcal on 26.1.) and goes until the end of treatment form. This is precisely the section of the CRF where the adverse event report is located. The second block of JPG contains all pages of the dose 3 visit audit log.
12261745 - one page in the audit trail of an adverse event of testicular cancer, detailing two confirmations that the subject had not initiated treatment on 9.10 and 13.11.
12312073 - this subject’s JPG page is the principal investigator’s signature on 16.3. by a Yanina Rivera, not Fernando Polack. There are two previous signatures on 21.10 and 22.1 with redacted names.
12313437 - this subject’s solitary JPG page is the confirmation of a positive local swab on 4.3. entered on 8.3. This subject had a very late revax contact on 22.3 and no record of a dose 3, nor related queries.
12315520 - this subject’s first JPG page is in the audit page of an adverse event of “Venous thrombosis of the right eye”, in which the site denies the presence of hospital admission covid testing; the other page is in the audit of an “arterial hypertension” adverse event containing Pfizer’s affirmative response 3 days after the site declares the adverse event was not sent to the safety database due to being “reported as a non-serious adverse event”.
12315632 - a page in the audit of deleted adverse event “Potential Covid-19 illness”, initially coded as pneumonia on 19.10., showing a 3.12 Pfizer query one month after the last edit demanding the site “harmonize reporting, and update in the appropriate location”, leading to the adverse event’s deletion on 13.12.
12461025 - this subject was discontinued after dose 1 due to an allergic reaction. of urticaria. The JPG page is in the audit of a second event of urticaria, which occurred after the first event had finished and was also marked “related”. The non-machine-readable page contains Pfizer’s queries about the causality assessment one week after entry: “Clin: Please confirm causality as urticaria post vaccination 1 had resolved,and this is a new event” as well as a query confirming subject discontinuation five hours after data entry: “CLINQUERY: Please clarify, has the subject been permanently discontinued from treatment? Thank you.”
12471135 - this subject has a block of ten JPG pages spanning the form comments (dated 1.10.) of visit 1 (30.9), all “not applicable”. This subject was discontinued after one dose due to adverse events of “Chronic cerebelar infarcts”, “Cerebral atrophy” and “Left Middle Cerebral Artery Infarct”, start dates 21.-23.10, no end dates, yet received two doses after unblinding, resulting in a fourth adverse event of injection site pain starting 17.2. with no end date. The infarct is related to “Pt.68 years old; and 50 pack year history of smoking as risk factors for event.”, but this is not added to the medical history.
These are the “full JPG” CRF files, where the entire contents are images instead of searchable text.
10911002 - 58 / F / placebo / BMI 30.8 / doses: 30.7 + 18.8 + 14.1 + 2.2
This subject had bilateral ear pain lasting six months starting 17 days after dose 1, and received doses 2, 3 and 4 while affected. The CRF is never re-signed by the PI after an edit on 2.2., with the last valid signature on 26.1.
10911170 - 57 / F / b2 / BMI 36.2 / doses: 25.8 + 16.9
The subject has an adverse event of related lymphadenopathy from 3.-30.9, so received her second dose while affected. Dose 1 was on 25.8. at 11:24, yet data entry begins on the 26.8., with several barcodes entered on the 27.8. The dose 2 visit data entry is also one day later, with the swab barcodes five days late. Visit 3 is entered five days late. The adverse event is entered a few minutes after the dose 2 data, and Pfizer fires off this query 35 hours later due to the investigator’s “related” judgement: “GDPCLIN: Per protocol an investigator’s causality assessment is the determination of whether there is a reasonable possibility that the IP caused or contributed to an AE. Please provide the facts or arguments to suggest a causal relationship.” The site gets around to answering “Related. Reactionary as result of receiving vaccine” 18 days later, on 6.10. The unblinding contact on 16.12. is entered on 11.1., invalidating the last signature from 2.11.
10911213 - 58 / M / b2 / BMI 29 / doses: 1.9 + 21.9
This subject’s 6.11. toxicity grade 4 myocardial infarction is not included nor mentioned at any point in the CRF, appearing only in the database. This means it was most likely not included in the 14.11. data lock interim clinical study report. The last valid investigator’s signature is from 12.11., and was invalidated by an edit on 24.2., which is when the unblinding ReVax contact on 17.2. was entered.
10971011 - 78 / M / b2 / BMI 31.3 / doses: 20.8 + 9.9
The covid illness has the “other” symptoms of generalized weakness, mild epigastric pain, mild dizziness, in addition to the protocol defined symptoms of cough and chills. The visit and swab occur one week after end of symptoms. Blood values from the ER visit show clear lymphopenia eleven days after the second dose. The adverse event of pneumonia lasts from 20.9 -5.10., while the covid illness ends on the 22.9. In the audit trail of the covid illness the following exchange is recorded from 12.-14.10.; Pfizer queries the delay of the swab, to which the site absurdly replies “Subject did not have COVID. Subject did have pneumonia. Chest X-ray on 10-5-2020 Showed Pneumonia resolved.” Pfizer: “DM1: Thanks for response. Please verify and confirm Date of Last Symptom resolved on the Signs and Symptoms form in the same visit. Else clarify” Site: “22Sep2020 is correct”. The dates are not changed, however. The only imaging results are “left upper lobe pneumonia” X-ray from 20.9. The site also tried to enter “no healthcare utilization” until Pfizer queries on the 16.10. (after the previous exchange): “Subject was seen in emergency room before hospitalization on 20Sep2020 for pneumonia and tested for SARs-COV-2. Please consider this healthecare utilization for potential COVID illness, and update as required.“
10971023 - 86 / F / b2 / BMI 29.3 / doses: 24.8 + 15.9
This subject was 86 years old (above the limit of 85) at time of enrolment and died on 21.12. The year of birth was initially entered as 1934, but the SAE report revealed the birth year was in fact 1933. After Pfizer helps the site along a little with basic maths, the error is corrected. The subject also has a deleted adverse event of gallbladder failure, which was in fact a gallbladder rupture, later deleted in favor of “septic shock” which was then also taken as cause of death. The fatal events began on 16.12. and are entered into the eCRF on 4.1.
10971064 - 78 / M / b2 / BMI 32.5 / doses: 31.8 + 24.9
There’s nothing immediately apparent as fraudulent in this subjects’ CRF; however, the site has to be prompted by Pfizer to enter the adverse events of “new onset atrial fibrillation” and “persistent atrial fibrillation” into the CRF. The first AE initially being coded as “shortness of breath” is troublesome, given it’s also one of the default covid illness symptoms. The event of “congestive heart failure” with the earliest start date of 22.12 is entered last, on 9.3. The subject also has an open-end covid illness with the same start date as Afib and shortness of breath as the only symptom, which only got swabbed one month after symptom start on 5.2. Pfizer comments on the delay, but as symptoms are still ongoing, the swab is protocol-compliant.
11101236 - 28 / F / b2 / BMI 24.9 / doses: 11.9 + 2.10
This subject has a highly suspicious covid illness starting the day after dose 2 and lasting until 9.10, the day of the illness visit on-site and central lab swab, with default symptoms diarrhea and chills and “other” symptom of “eye irritation”. The adverse event log contains “bilateral eye ichiness” and “bilateral eye redness”, but both adverse events are only from 14.-16.11. The adverse event of “rash to legs” was entered with no end date in the CRF, but there is an end date of 5.3. in the database. The adverse event of “throat itchiness” was queried for a covid test by Pfizer, but they were apparently satisfied by the site’s laconic response: “sub investigator determine not to do a illness visit”; the sponsor’s request for a unifying clinical diagnosis for the eye and throat AEs is similarly rejected. The audit also shows that the adverse event “change in vision” was initially reported as “change in vision, rule out TIA”, then as “transient ischemic attack”, until being changed to its final value on 26.2.
11231381 - 19 / F / b2 / BMI 32.5 / doses: 15.10 + 6.11
The reason this CRF was JPG’d is not immediately apparent. The only point to stand out is the last valid signature being on 20.1, invalidated by an edit on 16.2.
11351257 - 74 / M / b2 / BMI 29.4 / doses: 1.9 + 23.9
This subject has four separate events of atrial fibrillation, the first with start date UNK/November/2020 and ending on 9.12., the last starting 5.1. with no end date. There is also an adverse event of “stage IV non-small cell lung cancer” starting 16.11. with no end date, but you wouldn’t know any of this from reading the CRF, because the adverse event report form was not started. There are also no queries or reminders related to any of the adverse events to be found in the audit. To top things off, the last valid signature is from 26.10.
12411347 - 23 / F / placebo / BMI 23.4 / doses: 20.8 + 10.9 + 1.2 + 22.2
This subject has three additional adverse events of Fatigue, Headache, and Fever from 22.-23.2. which are not listed in the CRF. The subject’s only medical history item of dysmenorrhea is entered 63 days after enrolment, with no apparent prompt or query leading to the edit; the only other eCRF edit that day is the casebook signature form. The medical history has to be unlocked for changes to be made, and there usually is mention of this in the audit. The adverse event of related lymphadenopathy is queried for causality ten days after entry: “ClinQ: Please explain why the AE is related to the study vaccine”, to which the site replies “According to SI this event is rare but expected and is described in the Consent as “Lymph gland enlargement”. Notably, the last signature by designated site PI Edson Moreira is on 22.1., the later signatures on 4.2. and 24.2. have the PIs name redacted, but the redaction is too short for it to be Moreira.
44441634 - 43 / M / b2 / BMI 34.1 / doses: 23.9 + 14.10
This subject’s CRF is an absolute mess. There is only an adverse event of acute cholecystitis 28.12.-4.1. recorded, and a potential covid illness lasting from 28.12. to 31.12. The potential covid illness diagnosis is first entered as gastroenteritis and then changed to “stomach flu”. There are blood results from 29.12. entered in the covid illness showing astronomical liver markers which are not further commented anywhere in the CRF:
Alanine aminotransferase: 758 with a range of 14-63,
Aspartate Aminotransferase: 191 with a range of 15-37,
Alkaline Phosphatase: 205 with a range of 46 to 116,
Bilirubin: 3.5 with a range of 0.2-1.
The subject also has abnormally high leukocytes (12649, range 1530-7560) and a lymphocyte count of 0.01 which is clearly abnormal despite no provided reference range. There is also an abnormal abdomen ultrasound result from the same day: “Migrated in shape, size and conserved ecostructure. Non-dilated intra- and extrahepatic bile duct. Gallbladder with slightly thickened walls. (continue in comments)” + “with microlithiasic content, pancreas masked by meteorism, homogenous spleen of normal size”. The subject’s medical history is edited to include obesity ongoing since 1.3.2000 in November.
Suppression of female bleeding events
There is an auspicious lack of abnormal female bleeding events in the trial when compared to real world evidence. While the differences between process 1 used in the trials and process 2 electric bag-of-poo have been described broadly and I consider them a contributing factor to the dearth of bleeding events, there is evidence of systematic suppression of abnormal female bleeding. Here is how they did it: PIs were instructed to screen for gynecological AEs aggressively, so that bleeding events occurring during the trial could be attributed to the medical history and not even entered into the record.
It is noticeable that 2.203/22.497 female subjects have some form of gynecological medical history recorded which would be sufficient to act as a “cover”. 2.203 subjects with suspect bleeding MH history (IDList “AE”) of 48.091 have 20.873/197.037 MH items - that’s 4,58% of subjects with 10,59% of all medical history items. In contrast, there are 9.964 subjects with no medical history recorded.
54 of these subjects have CRFs. All gynecologically related medical histories were entered on day one, except for seven placebo patients: 10111029, relevant medical history entered one day after all other items, 10951009 added 12 days later, 11071044 added 56 days later, 11121122 added 169 days later, 12411347 added 63 days later, 12461131 added 110 days later, 44441297 added 51 days later.
The gynecological bleeding-specific MH screening is evidenced by the audits of two CRF files, 10071192 and 11291046. In both cases, the relevant medical history items initially include the terminology “excessive bleeding” which is queried out of existence.
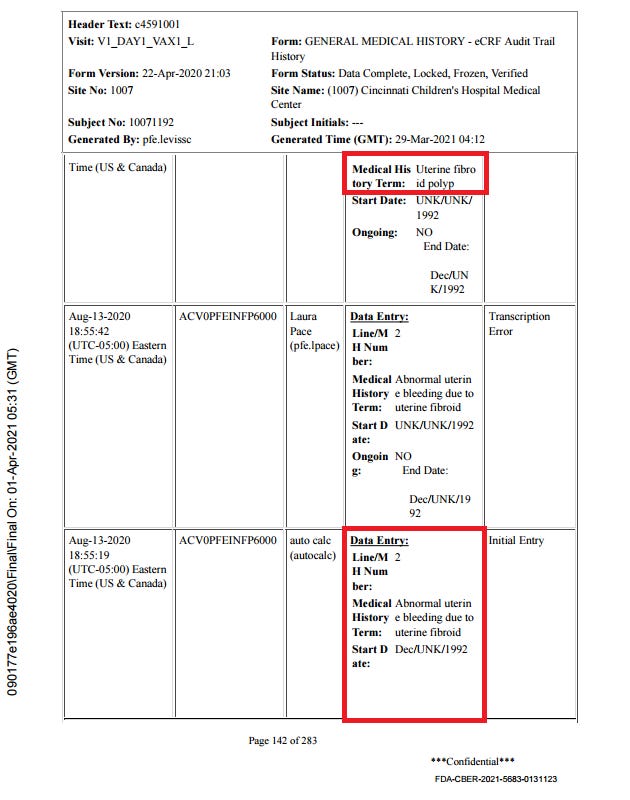
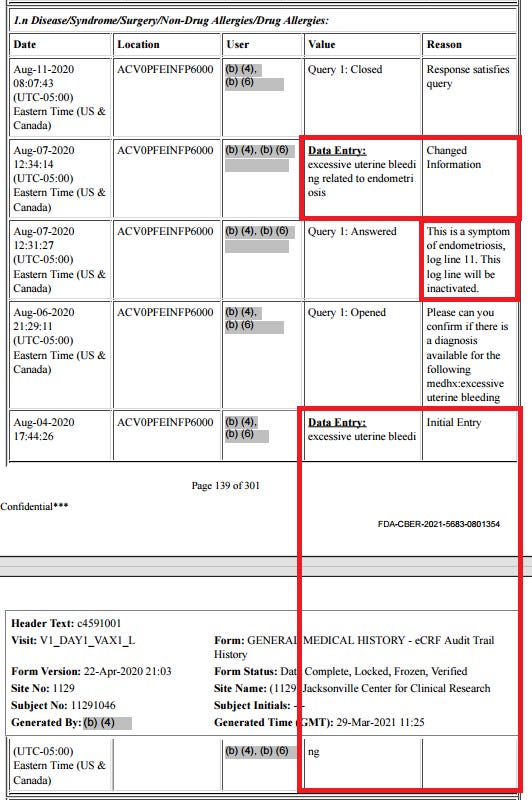
The spectacular diligence with regards to gynecological medical history is a central factor in the medical history theory of bleeding suppression: PIs were instructed in advance to screen diligently for gynecological issues, so that eventual bleeding-related disorders could be “automatically MH’d”.
The following 8 BNT CRF subjects have anomalies related to gynecological events indicative of suppressed bleeding events:
10791076 - a 59 years old female, BMI 36.2, received dose 1 on August 7, 2020 & dose 2 on August 28, 2020. Her CRF was included due to serious adverse event of uterine cancer, with a start date of January 25, 2021. The CRF reveals that the subject had a full hysterectomy & oophorectomy on February 8, 2021, where the start date is the day she scheduled her surgery, not the start date of corresponding symptoms. Pfizer repeatedly queried the site for reasons the subject scheduled the surgery, but never had an answer. It also is clear that there is no histopathologic diagnosis yet, as the site does not yet have the medical records. This subject obviously had abnormal bleeding, and it is very telling how studiously the site avoids mentioning this.
10841219 - 56/F/BMI 27.4, doses 1&2 17.8 - 9.9. CRF included for SAEs of uterine prolapse and left ovarian cyst, benign tumor. Both SAEs start on 24.8., one week after dose 1, and conclude on 19.9., so the subject received her second dose while these events were ongoing. On page 51, the uterine prolapse is claimed to have occurred previously, however the medical history only has a bladder prolapse. Again, how is an ovarian cyst diagnosed in a post-menopausal woman? Probably because she started bleeding, dont you think?
10851018 - 40/F/BMI 36, doses 1&2 1.-22.8. CRF included for SAEs of endometriosis and endometrial thickening, 9.-11.2. Clearly a bleeding related adverse event, the reason this CRF stands out is that the AE is not logged, despite Pfizer asking the site to do so.
12311315 - 41/F/BMI 25.1, doses 1&2 15.8.-3.9. CRF included due to SAE of vaginal malignant melanoma and anemia. The anemia adverse event has the causality assessment of “probable relationship with vaginal tumor under study”. How might anemia be related to a vaginal tumor, perhaps due to bleeding? A clear-cut example of direct mention of vaginal bleeding being studiously avoided.
12313028 - 30/F/BMI 21.5, doses 1&2 on 21.8. - 11.9. CRF included due to adverse events of special interest (AESI) lymphadenopathy and SAE of opticus neuritis. The AE log also shows an adverse event of “metrorrhagia” 13.-19.9., which means bleeding in between periods. The CRF has no further explanation. They couldn’t just say dysmenorrhea, they deliberately chose a softer term.
12314690 - 60/F/BMI 21, doses 1&2 28.8.-16.9. CRF included for SAE of signet ring gastric carcinoma. The subject initially had an adverse event of exacerbation of gastroesophageal reflux disease, which led to a first update of the medical history with GERD on 30.10. However, a second update occured on 29.3. which included a hiatal hernia, initially with end date of 17.11.2020, changed to be ongoing, and the items “uterine myoma” and “left ovarian cyst”, both with start date July 2019, both ongoing. Were these medical history items not entered initially or in October because they only became necessary when there were adverse events to keep out of the log, namely hernia surgery and abnormal vaginal bleeding? It is also notable these medical history items are not in the database, as they were added to the CRF on 29.3.
12315404 - 79/F/BMI 36, doses 1&2 30.8. - 8.10. Dose 2 was delayed due to an “uncomplicated urinary tract infection” lasting from 31.8.-2.10. CRF included due to SAE of “High grade infiltrating urothelial carcinoma”. Also in the AE logs is “exacerbation of gross hematuria” from 11.10.-29.12, which means blood in urine, and lots of it. The CRF reveals that the “uncomplicated UTI” was initially hematuria as well, until it was changed by a redacted user on 5.1. They waited for the subject to stop bleeding and vaxed her again, and when she developed a serious cancer, they fraudulently changed the AE term of the initial bleeding disorder. The stop date coincides with the date of cancer diagnosis, showing that these AEs are routinely hidden in overarching coding terms.
12411051 - 33/F/BMI 32.5, doses 1&2 12.8.-2.9. CRF included for AESI of lymphadenopathy 3.-6.9. Also in the AE logs is an adverse event of “cystitis”, from 7-14.10., related to “bacterial urinary infection”. Along with the medical history of ongoing polycystic ovarian syndrome, the choice of cystitis as AE term instead of UTI makes this subject a suspect for unreported abnormal bleeding.
When looking at the possibly bleeding related adverse events amongst the CRFs, there is a clear coding disparity in favor of BNT. The database has 69 definite-to-possible female bleeding adverse events, 23 of which are in CRFs, 7 BNT - 16 placebo.
12411385, the only CRF subject with a specific coding of “dysmennorhea”, is in the placebo arm and the adverse event lasts one day on 5.9. and is related to “menstrual period” - this is absolutely not vaccine-induced dysmennorhea as described widely in real life, which is characterized by duration, severity, and anachronicity.
Parallel IT systems
In the context of this study, the CRF is more like a collage, pasted together from different data sources to create an optimized deliverable for regulatory submission. The Clinical Study Data Reviewer’s Guide51 and the “Annotated Study Book”52 (a sample CRF form showing where database variables are pulled from) explain the technical side; what isn’t mentioned is the use of up to 6 parallel IT infrastructures for sharing of medical records, source data, blinding status etc, including Florence, Redcap, Impala, Argus, Trialmax and Complion53, or that sites kept physical paper binders as source documentation.54
Florence
Source file, progress note, and subject status sharing platform.
11301031 Page 153 - regarding covid symptoms reported by the subject but not given a covid visit by the site: “Symptoms not deemed Covid-19 symptoms during conversation with patient. Related to dramatically reduced air quality d/t extensive fires. Note uploaded to Florence”
12511050 Page 337 - answering a query why vaccination 2 was not given: “SUBJECT NOT TO RECEIVE VACCINATION PER PI DUE TO CHRONIC HEPATITIS C. PLEASE READ PROGRESS NOTE IN FLORENCE.”
10681091 Page 196 - a query and answer on the Further Vaccination Confirmation page: “unable to verify please upload completed Form # 1” - “in Florence, under source folder titled "Form1"
Redcap
Adverse event/health outcome monitoring system.
10301059 Page 143 - In the audit log of a right lung adenocarcinoma: “For AE Right upper lobectomy of lung: Response to "What was the outcome of this adverse event?" is 'Unknown' but End Date/Time is provided or "Is the adverse event still ongoing?" is marked "Yes".” - “will update in redcap to not still going on.”
12312914 Page 317 - In a covid visit that was ongoing while a new one had already been entered: “CRA: Please verify: as per medical chart, symptoms for this COvIll ere not documented as solved. Correct accordingly. Thanks.” - “As per information loaded in RedCap, this information is correct. Thank you”
Impala
Subject information system including blinding
11311222 Page 83 - some confusion about the subject’s gender in the audit of visit one: “As per Impala, Sex is recorded as Male. Please review and update as appropriate.” - “Subject is female; we will review Impala and correct as appropriate. Thank you.”
10441093 Page 147 - the site didn’t update blind status appropriately “IVRS Reconciliation: Subject Safety Concern is selected as the primary reason for un-blinding but matching blind break is not captured in IRT. Please correct primary reason in Inform or confirm as correct”. - “Subjects unblinded for TE safety concern at v1 or v2 vax and not PA10. Sponsor requested subjects unblinded in Impala on 23/24Nov2020 (prior to PA10) and unblinding explained to each. unblinding not done in Impala/IRT which has resulted in a IRT mismatch”
Argus
Brook Jackson shared a screenshot of the Argus interface on twitter55, highlighting the considerable redundancy between systems. In C4591001, Argus seems to be related only to serious adverse events.
11141006 Page 187 - in the adverse event audit of a pulmonary embolism related to blood coagulation disorder: “clin: requested info not seen in Argus. Pls provide start date of coag disorder. If prior to enrollment should be added to MedHx CRF, if after pls report as separate AE. Thanks”
10811026 page 418 - “Ute Frantz” authors a query informing the site the adverse event “Neoplasm of uncertain behavior of left ovary” was still listed as “LARGE PELVIC MASS” in the safety database: “Left query open until ARGUS gets updated as of 10/23/2020 not updated yet.”
Trialmax/trialmanager
This eDiary software by company Signant Health was used for reactogenicity and covid illness apps. There are also a few mentions of Signant itself.
11281267 Page 150 - an error in reactogenicity subset assignment reveals that reactogenicity subjects were apparently not assigned by chance: “Email sent for CRA/Signant notification on 28Sep2020.” - “subject was inadvertently entered into reactogenicity subset in error, on the trialmax system. EDC and source are correct.”
1261037 p185 - a very common exchange betwenn Pfizer and the site regarding a patient “illness” eDiary entry: “RQ1: Thank you for response. Per protocol, COVID Illness Symptoms should only be reported on the COVID Illness form and not on the AE form. Please update or clarify.” - “it isn't a covid illness-the patient reported his hospitalization in the trialmax app but it was his SAE symptoms which resulted in surgery”
Complion
Seemingly another source file sharing platform/database.
10951101 Page 254 - a Pfizer query in the audit of a Hepatitis B medical history item: “Please provide the source in Complion under Medical History, thanks”
10811135 Page 175 - After the site made a covid visit erroneous on 31.10: “on 30Oct2020 an InFOrm query response states "AE will be entered as this has now turned into an SAE. subject was hospitalized". There is no SAE reported and no source in Complion. Please report SAE This pot covid visit took place and will need a conv vis”
Thanks to @ReportAdve30828 on twitter, I just recently found a Ventavia QA/QC study summary report which also references Complion and Impala. There is a lot to unpack in that letter which will take some time, but it was too important not to share. It was made public by Brook Jackson via Paul Thacker’s Disinformation Chronicle.
Enrolled after 14.11.
Mr. Kremer (from the introduction) tipped me off to a clear manipulation of the study population. There are 2.826 / 48.091 subjects enrolled after the data cutoff point of 14.11. for EUA. 2.202 belong to the adolescent 12-15 population, leaving 624 in the 16+ population. Closer inspection reveals that Pfizer took the 16+ very closely to heart. All 624 subjects together have an average age of 23.2 years, but when excluding sites 1218, 1219, 1220 and 1221, there remain 479 subjects with an average age of just 16.5, precisely seven of which are not 16 or 17 years old56, 5 are eighteen, 1 is twenty, and one subject is 68 years old. In contrast, the “oldie” sites 1218-1221 have an average age of 45.5 across 145 subjects, but when disregarding these sites and the seven exceptions, Pfizer enrolled only 16 and 17 year olds after the efficacy endpoint had been reached. Clearly a premeditated decision, perhaps due to knowledge gained about age as a risk factor for adverse events? This also ties in with the mandatory reactogenicity subset assignment in this age group, directly enabling adverse event obfuscation.
Missing nucleocapsid antibody and PCR results
Several database “issues” have become apparent in the course of the CRF review. There is precisely one barcode entered into the MB (microbiology) database for the dose 3 & 4 swabs of placebo patients being unblinded.57 There are 35.825 dose 3 & 4 visits listed in SV.58 Collection of a PCR swab pre-dose was part of the Schedule of Activities; it is unclear why this data was not entered into the database, as the available CRFs show recording of these barcodes. This extra swab data would be a nice data point to have considering the large amount of SAEs in placebo patients who received their unblinding vaccine doses, which might be why they aren’t supplied.
Another issue with SV is the extremely sporadic presence of a value in the SVREFID column, with the description of “Reference ID”. The possible values besides none are CLINIC VISIT, TELEHEALTH VISIT, and TELEPHONE VISIT, data which was recorded in the CRF, often queried by Pfizer, and often denoted in the CO database file.
There are 82 N-binding antibody serology results from covid convalescent visits entered into the IS (immunogenicity) database.59 There are 8.693 16+ covid convalescent visits in SV(VISIT=COVID_A1-F1) for 7639 subjects, and the CO database contains confirmation of serum samples collected during a covid convalescent visits for a minimum of 7506 16+ subjects (RDOMAIN=IS, VISIT=COVID_A1-F1, COVAL=Y, “minimum” because many non-Y “Sample collected?” responses are positive for serum collection but denote an administrative change or a blood draw complication). The damning extra tidbit of information? Until protocol amendment 6, which changed all blood draw volumes to 20 mL, convalescent visits had twice the amount of blood drawn with 50 mL than all other study visits with 25 mL, probably due to extra retention samples kept on-site. In other words, the immunogenicity results for more than 8600 covid convalescent blood draws are missing; amendment 6 came into force on 8.9.2020, by which time precisely 10 convalescent visits had occurred, so approximately 172,5 liters of blood were drawn with no data provided. As someone who draws blood every weekday, 50 mL is an absurd amount of blood - a standard draw consists of one serum vial with 8 mL and one EDTA vial with 3 mL. I am not aware of justifications for this missing data.
That this data was apparently not entered into the database is suspicious, given the insights regarding vacinee failure to generate nucleocapsid antibodies when infected gained from Moderna’s trial60 and inexplicable, as Pfizer had delayed the analysis of crucial human samples from BioNTech’s first-in-human study for about a year, citing priority of work on two non-C4591001 studies, tacitly implying the workup of C4591001 samples had concluded.61
Covid illness data quality
Covid visits are recorded in different databases, these counts however do not match up. In database Subject Visits, there are 9.055 unique subjects with covid_a illnesses, in database Analysis Dataset SYMPToms, there are 9.005 records of “chills” associated with a covid_a illness, in Clinical Events there are 8.549 “COVID-19 like illness? Y” rows associated with covid_a visits, finally, there are only 7.927 covid_a central lab swabs in the MB (microbiology) database.
There are 7.503 subjects with a covid_a entry in all four of SV/ADSYMPT/CE/MB, including 310 of 372 CRF subjects with a covid_a illness.
In CE, where toxicity grades and diagnoses for potential covid illnesses are stored, the breakdown of CRF/non-CRF cases is as follows:
100% of tox 5 (7/7 unique subjects)
83% of tox 4 (15/18 unique subjects 12311531 12313895 12311087 not present)
35,4% of tox 3 (45/127 unique subjects)
7,3% of tox 2 (57/781 unique subjects)
5,6% of tox 1 (86/1.521 unique subjects)
0% of tox 0 (0/3 unique subjects)
3,3% of no tox grade assessment (224/6.682 unique subjects)
There only being 3 subjects with toxicity grade 0 in CE is simply Pfizer’s incredibly piecemeal data point completion; 10471290, 10971061 and 12481120 are CRF subjects with “asymptomatic” covid illnesses where there just wasn’t a toxicity grade entered into the database.
Reactogenicity
CE also contains the reactogenicity eDiary records. The breakdown according to CRF/non-CRF is as follows:
Potentially Life-Threatening
0/5 subjects with 0/5 potentially life-threatening events62
Severe
27/646 subjects (4,17%) with 46/1.023 (4,49%) severe reactogenicity events
Moderate
126/4.562 subjects (2,76%) with 317/12.558 (2,52%) moderate reactogenicity events
Mild
172/ 7.081 (2,42%) subjects with 484 /21.061 (2,29%) mild events
Y without grading
32/1.353 (2,36%) subjects with 44/1.916 (2,29%) events that occurred without receiving a toxicity grading.
Neither Y or N
8/236 subjects (3,38%) with 77/2.772 (2,77%) “unknown if occurred” or perhaps “legally could not enter N but an entry of Y would be inopportune” events
No reactogenicity events
Out of 257 CRF subjects in the reactogenicity subset, a total of 66 (25,6%) made no affirmative entries in their reactogenicity eDiaries, compared to 2.556/10.275 (24,8%) amongst all reactogenicity subjects. These 66 “clean” reactogenicity CRFs consist of 11 BNT and 55 placebo subjects, including 2 and 3 subjects who died, respectively.
Of 38 subjects that died, 13 were in the reactogenicity subset (~34%), compared to 22% of the 16+ study population (10.275 / 45.786), but this difference does not achieve statistical significance (calculation below courtesy of OpenVAET).
As is evident from the breakdown by severity, the worst tiers of reactogenicity events are severely underrepresented amongst CRF subjects compared to adverse events and potential covid illnesses. This is especially pertinent due to mandatory reactogenicity subset enrollment for all 12-15 and 16-17 subjects; a very handy way to record adverse events without having to supply a CRF file.
Another interesting point is what I call the CE duration glitch. Reactogenicity events in CE have a CEDUR column, which according to the .xpt file means “Duration”. It actually denotes how many days inside the seven-day reactogenicity window the event lasted, so it can only have values between 1 and 7. The actual duration of the event can be easily calculated by subtracting CEENDY from CESTDY, which are “Study Day of End of Observation” and “Study Day of Start of Observation” respectively; despite these variables being supplied, the database has to be modified to include the “true” duration values. Doing so reveals that out of 46.095 reactogenicity events with calculable duration, 402 have a “true” duration greater than 7 days,63 and 139 greater than 14 days, with the top 5 being 69 days of severe injection site pain (10901043), 68 days of moderate fatigue (11351048), 41 days of mild injection site pain (11291261), 38 days of mild diarrhea(11351129), and 37 days of moderate fatigue (11141024). None of these subjects having corresponding adverse events logged in flagrant violation of the protocol; with the exception of 11351048, who has dose 3 & 4 adverse events recorded to which reactogenicity subset doesn’t apply, none of these subjects have any adverse events recorded at all. To pick up the initial theme, the CEDUR values are 7 for the first three events and 6 for the other two. 11141024 from the listing above also has an event of mild diarrhea lasting 32 days with CEDUR=1, because it started on 10.9., six days after dose 2 on 4.9.
The CRF subject with the longest reactogenicity events is 12171044 with mild chills and moderate muscle pain from 10.11. to 10.12. after receiving dose 1 placebo on 4.11. The only adverse event for this subject is anxiety with a start date of 21.11. and lasting til 4.3.; the subject also had a positive covid illness and nearly died in hospital, despite no recorded medical history. Whether this subject really did receive placebo is questionable due to lab results from the hospitalisation showing clear lymphopenia 1 week post-dose. The subjects’ “anxiety” ends on 4.3., the same date he withdraws consent. The corresponding form has the memorable quote “[He withdrew from the study because he give up vaccinated.]” Better late than never! It’s also important to note this subject was initially entered as BNT trial arm assignment on the CRF, which was only changed to placebo 4 days later.
There are 73 reactogenicity events with no end date in 57 subjects.64 None of the five CRF subjects with open-end reactogenicity events have corresponding adverse events entered.
10161087 - 43 / F / b2 / BMI 24.4 / dose: 8.10
Severe headache starting day of dose 1, recorded as ongoing 31.8., never queried for completion. This subject also had a 25x13cm injection site swelling which still didn’t manage to meet Grade 4 criteria with end date 21.8. There is a noteworthy exchange between Pfizer and the site on page 228. It’s unmistakeably the case that the headache should be on the adverse event log, and that the site intentionally misconstrued the protocol in order to avoid this, even against Pfizer’s direct instructions. Pfizer in turn failed to press the issue for the next ~8 months of study and neglected to adress the issue post-facto, as they did with numerous other files.
10811026 - as covered previously, the reactogenicity “pool” was abused to keep the adverse event of diarrhea off the adverse event record. The corresponding covid illness visit has an end date for the diarrhea of 5.10., and the reactogenicity eDiary assessment form shows no queries after the initial “ongoing” entry on 1.10.
11201350 - 67 / M / b2 / BMI 31.1 / doses: 20.10 + 20.1
Moderate fatigue and joint pain starting day after dose 2, 20.1. The corresponding vaccination symptoms CRF form was initially completed as “NO” to all answers on 30.12., presumably due to the delay between doses, then to the final state “Not Done” on 11.1., showing that Pfizer very much had the capacity to retroactively change data entry points. There are entries as late as March, so the reason the reactogenicity eDiary assessment remained undone is unclear, but the site had to be asked repeatedly to even enter the second dose and 1 month visit when they did occur, hinting at deeper problems in the data quality workflow.
12171044 - 34 / M / placebo / BMI 28.3 / dose: 4.11
Not only the longest-duration reactogenicity events amongst CRF subjects, but an open-end one, too: mild fatigue starting 10.11., six days after vaccination. Initially entered with a stop date of 30.11., this was changed to “ongoing” five days later on 15.12. and never queried for completion.
12461025 - 45 / F / b2 / BMI 21 / dose: 28.9
Severe fatigue and headache starting two days after study start entered as ongoing on 19.10., never queried for completion. The subject’s 8 other reactogenicity events were all over by 29.-30.9.
What’s the purpose of all these fancy IT systems with source sharing when none of these events end up being recorded as adverse events?
The protocol amendments
Being familiar with the study protocol proved to only be a starting point for a formal investigation of changes to reporting guidelines. Notably, there is only a “tracked” version of the protocol which lists displays changes word-for-word available for amendment 18 in the PHMPT Pfizer 12-15 production. All other versions of the C4591001 protocol released by the FDA are simply consecutive amendments tacked onto each other - in contrast, BNT162-01’s protocol includes a tracked changelog starting on Page 150. Splitting the available interim-mth6 protocol file into separate amendments enabled comparison using tools like DiffPDF.65
There is a “hidden” amendment #15 which came into effect on 25.3.2021, after the declared cutoff of 13.3., yet inside the timeframe of the “database snapshot” on 25.3.66 and CRF finalisation and submission starting 29.3. and lasting into April, but is not included in the 16+ protocol. It is included here for completeness sake.
There are several articles worth of information only related to the protocol and its changes, so this section will provide a brief overview of the amendments and focus only on the outcome-reporting related issues.
PA 0 (Original protocol) - 15.04.2020
The first C4591001 subjects were enrolled on 29.4, and injections began on 4.5., so this version was valid for the first nine days of application to humans. It remarkably opens with the sentence “This is a Phase 1/2, randomized, placebo-controlled, observer-blind, dose-finding, and vaccine candidate–selection study in healthy adults.”, only to state “The study consists of 3 stages.“ three paragraphs later. This is connected to the sponsors’ initial intentions of running the Phase 2/3 trial separately under C4591002.67
PA 1 - 13.05.2020
Perhaps the most substantial change right at the beginning: “Clarified that efficacy against COVID-19 is based upon illness (not infection) rate ratio”
Interesting to note are the changes of serum neutralisation from SARS-CoV-2 wild-type to just SARS-CoV-2 and from “spike protein-specific” to “S1-specific” antibodies.
PA 2 - 27.05.2020
Adjustment of safety criteria for dosing cohort progression, changes to haemoglobin safety data assessment
PA 3 - 10.06.2020
This amendment came into force on the same day of the Type C meeting between FDA and Pfizer, in which Pfizer stated their intent to run Phase 3 with BNT162b2 30 ug. The amendment introduces illness eDiaries, increases the number of doses possible per vial, extends the Phase 1 screening window, and removes BNT162a1, BNT162c2 and BNT162b3 from the study
PA 4 - 30.06.2020
Efficacy objectives formally added to the study, BNT162b3 is added back into the protocol, illness visits possible per telehealth or in-person, covid symptoms updated to align with FDA-accepted definitions, AE reporting adjustments.
PA 5 - 24.07.2020
The Phase 3 protocol amendment, dosing starts three days later. BNT162b3 is kicked back out, changes to covid illness/adverse event reporting guidelines, immunogenicity becomes exploratory, and clarification that while all subjects will have blood drawn, only certain subsets will be analysed, “3-tier approach to summarizing AEs”
This protocol amendment includes a crucial change to protocol section 8.3.7, “Disease-Related Events and/or Disease-Related Outcomes Not Qualifying as AEs
or SAEs”. Previously reading only “Not applicable.”, PA 5 inserts the following fantastical paragraph:
Potential COVID-19 illness will not be reported according to the standard process for expedited reporting of SAEs, even though the event may meet the definition of an SAE. These events will be recorded on the COVID-19 illness pages in the participant’s CRF within 1 day.Potential COVID-19 illness events will be reviewed by a group of internal blinded case reviewers. Any SAE that is determined by the internal case reviewers NOT to meet endpoint criteria is reported back to the investigator site of incidence. The investigator must report the SAE to Pfizer Safety within 24 hours of being made aware that the SAE did not meet endpoint criteria. The investigator’s SAE awareness date is the date on which the investigator site of incidence receives the SAE back from the internal case reviewers.
PA 6 - 08.09.2020
SoA rearranged, blood draw volume reduction, upper and lower age limit changes, sample size increase, and the infamous “Added clarification that potential COVID-19 illnesses that are consistent with the clinical endpoint definition should not be recorded as AEs.” Reminder: exactly 10 covid convalescent visits had occurred at this time
This amendment introduces the following critical phrasing in several parts of protocol section 8: “Note: Potential COVID-19 illnesses that are consistent with the clinical endpoint definition should not be recorded as AEs. These data will be captured as efficacy assessment data only on the relevant pages of the CRF, as these are expected endpoints.”
PA 7 - 06.10.2020
Symptoms within 4 days of a potential covid illness are to be counted as an extension of the existing illness, changes to 7-day-post-dose swab embargo, change to testing guidelines /wrt repeat tests, Section 8.15 added, scope of safety analysis adjusted
PA 8 - 15.10.2020
Repeat-covid surveillance “clarified” to state that having been positive doesn’t mean end of surveillance for potential covid illness, and “clarification” that reactogenicity in non-reactogenicity subjects should be recorded as an AE; conversely implying that reactogenicity events for reactogenicity subjects should not be recorded as AEs, contradicting the guidance that reactogenicity events longer than 7 days should be recorded as AEs
PA 9 - 29.10.2020
No longer content with excluding covid cases 1-week post-dose 2 in analysis, the ultimately utilized efficacy window of 14 days post-dose 2 is introduced, purportedly “To better align with the natural history of SARS-CoV-2 infection”, mandatory reactogenicity subset assignment for 16-17 year-old subjects from this point onwards
PA 10 - 01.12.2020
The “unblinding” amendment came into effect on the same day as UK’s Regulation 174 authorization, possibility to unblind both according to local recs and protocol, antibody titres above an upper bound excluded from analysis, additional telehealth provisions
PA 11 - 04.01.2021
Asymptomatic surveillance period schedule of activites added to protocol, in which certain subjects conduct biweekly swabs regardless of symptoms
PA 12 - 14.01.2021
Asymptomatic surveillance only in blinded patients, confusing statement regarding N-binding antibody blood draws, “Corrected the exploratory objective to describe non-S seroconversion to SARS-CoV-2 to clarify that this will only include participants who received BNT162b2 at initial randomization (since those who received it subsequently do not have blood drawn).”, modification of exclusion criteria #5 so covid-positivity is no longer grounds for discontinuation of treatment
PA 13 - 12.02.2021
Phase 1 booster amendment, reactogenicity population adjustment, PI discretion for unblinding doses now advised, unlinded asymptomatic surveillance subject should continue swabbing regardless
PA 14 - 02.03.2021
Guidance that “emergency” unblinding is to occur outside of IRT, asymptomatic case definitions added, “clarified” that swabs and blood draws can be used for multiple visits if these visits coincide, additional variant + BNT162b2-naive booster trial arms introduced
PA 15 - 25.03.2021
V101/102 permits administration of vaccine to pregnant subjects in some cases and EDP reporting requirements reduced to 28 days post-dose
Crucial protocol sections and when they changed
Protocol section 8.2.2. “Electronic Diary”
This section is changed in PA 4, with reactogenicity no longer being solicited from all subjects, to Phase 1 + 2 and “at least the first 6000 randomized” only. HIV+ (PA 6), 12-15 (PA 7) and 16-17 (PA 9) subjects are added in later amendments, but otherwise the wording is changed insubstantially.
Protocol section 8.3.7. “Disease-Related Events and/or Disease-Related Outcomes Not Qualifying as AEs or SAEs”
Notably added already in protocol amendment 4 on 30.6.2020, stating only “Not applicable.”, the text quoted above was added in PA 5. The text was changed in PA 668 to specifically emphasize the categorization fraud, expand the scope to “potential COVID-19 illnesses and their sequelae”, and specify a timeframe of 24h in which the covid illness CRF form should be filled out, which was done extremely rarely. The text was changed again in PA 7 on 6.10.2020 to include the possibility of SAE reporting at investigator’s discretion.69 The only change after that was a switch in section to 8.3.8. in PA 13.
Protocol section 8.13. “COVID-19 Surveillance (All Participants)”
The first change is introduced with PA 3, making mention of illness eDiary use. This is also the amendment which added section 8.14., which details the use of third-party apps and devices. PA 4 implemented several important changes: the phrasing was changed to include the possibility of an in-person visit; previously the wording was exclusively telehealth, the sentence “During the 7 days following each vaccination, potential COVID-19 symptoms that overlap with solicited systemic events (ie, fever, chills, new or increased muscle pain, diarrhea, vomiting) should not trigger a potential COVID-19 illness visit unless, in the investigator’s opinion, the clinical picture is more indicative of a possible COVID-19 illness than vaccine reactogenicity” is included, and the list of covid symptoms is changed to covid diagnosis, fever, cough, shortness of breath, chills, muscle pain, loss of taste/smell, sore throat, diarrhea, and vomiting. Wheezing, sputum production, and nasal congestion/discharge are dropped from the protocol listing yet remain in the database list of symptoms. The next change occurs with PA 6, with the qualifier “irrespective of perceived etiology or clinical significance” added to when a potential covid illness visit should occur, and adds the biologically absurd “4 days after symptoms end” swab deadline. PA 7 adds the “symptoms within 4 days are one covid illness” guidance and adds another “investigator’s discretion” qualifier to the 7-days-post-dose testing embargo. The last change is with PA 8 nine days later, which adds a reminder that “Surveillance of potential COVID-19 symptoms should continue even if a participant has a positive SARS-CoV-2 test earlier in the study.”
Protocol section 10.3. Appendix 3: Adverse Events: Definitions and Procedures for Recording, Evaluating, Follow-up, and Reporting
This section defines AEs/SAEs, clarifies what does and does not constitute an adverse event and how these should be recorded and reported, covers intensity and causality assessments and follow-up guidance. The major loopholes of medical history with the necessary worsening/exacerbation qualifier, along with the “disease being studied and its progression are not AEs”, are codified from the start. The only non-editorial changes to this section were implemented with PA 4: the exposure during pregnancy CRF reporting guidelines are changed from “None” to “All AEs/SAEs associated with exposure during pregnancy or breastfeeding” and a paragraph stating that “Suspected transmission via a Pfizer product of an infectious agent, pathogenic or nonpathogenic, is considered serious”.70
The absolute majority of references to the protocol are made by Pfizer when bullying a site about “miscategorized symptoms of disease”. Beyond that, it quickly becomes apparent the protocol was little more than window dressing, utilized to bludgeon recalcitrant sites into compliance and ignored when opportune. The loopholes used to systematically confound and fraudulently manipulate the data were present from the onset of Phase 3; the only actual changes were Pfizer having to repeatedly spell out and clarify what was expected of sites and their principal investigators due to being fundamentally counterintuitive.
Deviations
This is an area which still needs more scrutiny and analysis. Beyond some preliminary analysis related to covid visits, I haven’t really dug into these yet. Some observations are that sites seemingly were responsible for recording protocol deviations, at least partially, and that a large proportion of deviations are not captured in the CRFs, which contributes to not knowing what they mean; the nebulous formulations don’t help.
Amongst CRF subjects, there are 65 unique deviation terms. Amongst non-CRF subjects, there are 107 unique deviation terms. Amongst all subjects, there are 109 unique deviation terms. In other words, there are two deviations only present in CRF subjects. The two deviations in question are “Participant failed to meet inclusion criterion #01 (Male or female participants between the ages of 18 and 55 years, inclusive, 65 and 85 years, inclusive, or 18 and 85 years, inclusive, at randomization (dependent upon study phase).” and “SAE not reported to IRB per local regulations”. The age deviation is logged for 10971023 (86 years old, covered in the JPG subject section) and bizarrely 11421084, 42 years old. The IRB SAE reporting deviation is logged for 12312576, 24/F/placebo. Both subjects deserve a brief narrative.
11421084 - 42 / M / placebo / BMI 28.3 / doses: 12.8 + 4.9
This subject was marked ineligible for not disclosing medical history of major depression and suicidal ideation on 30.11. The toxicity grade 4 adverse event of suicidal ideation from 19-21.8 was entered into the CRF on 15.9, so the subject received the second dose after this event occurred. There is a cryptic concurrent hospitalization for an “altercation”, later changed to injury to right orbit, then to closed head injury. There is an excellent site query dated 29.10. in a discussion in the inclusion/exclusion criteria audit: “I requested guidance about this subject a month ago and have not yet received word about what to do with him.“ The subject’s only other protocol deviation is “SAE report delayed or not reported to Sponsor”, making the age deviation seem like a mistake.
12312576 - 24 / F / placebo / BMI 23.2 / doses: 20.8 + 9.9 + 27.1 + 18.2
Despite having no medical history recorded, this subject first had a renal colic on 20.10, then had both gallstones (6.-8.11.), kidney stones (5.11.-21.12) and right hydronephrosis (5.11. - open end), and injection site erythema for one day after dose 4. The gallstones were discovered as an incidental, asymptomatic finding during the workup of the kidney stone adverse event, which started as a second instance of “renal colic”. The kidney stones were treated with something called a double-J stent, which was only added after a Pfizer prompt one month after data entry: “CLINICAL SAE report states a double-J stent was placed for the event Nephrolithiasis; please review if this is accurate and a Non-Drug treatment was given.”, this only occurred after the November data cut-off. In the audit of the renal colic event, a site data entry shows the subject was asymptomatic between the events: “I speak by phone with the volunteer who says that she still does not have a turn to carry out imaging studies. She is asymptomatic.” Pfizer confirms the lack of medical history in several different queries.
There are 13 subjects71 older than 85 enrolled in the trial besides 10971023. 5x 86 years old, 3 BNT, 1 placebo and 1 screen failure, 3x 88 years old, all placebo, 2x 89, both BNT, and two 91-year-old placebo subjects. Only 10971023 has an age deviation, and is also the only subject that died, and the only 85+ to receive a CRF.
Results of scraping the CRFs for the keywords “deviation” and “PD tracker” and cross-checking with subjects who have no deviations recorded: a list of fifteen subjects, two of which have “nasal septum deviation” as an adverse event respective medical history item. Further excluding one subject where a protocol deviation was threatened regarding an erroneously entered dose 2 date, and two subjects with visits 1 and 4 days out of window, all remaining ten subjects are instances of data being changed to avoid deviations being logged:
10551176 - wording is changed from “subject received non-study coronvirus vaccine” to “subject withdrew consent prior to receiving non-study coronavirus vaccine” in order to avoid having a deviation logged. The discontinuation is recorded on 22.12, the non-study vaccination form updated by 14.1, and the phrasing changed by 22.2.
11111109 - subject status changed from “adverse event” to “subject withdrew consent” to avoid having a deviation logged for missing dose 2
11341153 - potential covid visit “not done” deviation avoided by marking adverse event causality as “related”
11691055 - dose 4 pregnancy test was initially “Not done”, populated with data after a Pfizer query demanding result or deviation
11781300 - potential covid visit “not done” deviation avoided by entering adverse events into CRF as “related” after Pfizer queries subject eDiary symptom reports
12313345 - the audit of a bladder carcinoma adverse event shows the subject experienced a number of potential covid symptoms that were never swabbed, and a protocol deviation was agreed to be logged but never was: ”Clin: Thank you. A protocol deviation will be documented.”
12314197 + 12315186 - both subjects have deviations threatened for missing covid convalescent visits
12315559 - adverse event causality is changed to “related” in order to avoid a “swab not done” deviation
12641229 - due to “Subject was currently traveling and did not bring his swab kit, swab could not be collected until his return home and discharge from hospita”, the covid illness is missing swab barcodes until 17.12.; a deviation for the swab being out-of-window is avoided due to the symptoms initially ending on 8.12. being extended to “ongoing? yes” on 18.12., and changed to stop date 15.12. one month later.
There is an oddity of the Analysis Dataset Deviations (ADDV) which warrants closer inspection - a free-text data point describing the visit a deviation is associated with, abbreviation “DESGTOR” with descriptor “Visit Designator” in the xpt file. There are 197 unique terms amongst the 37113 individual entries, including 32 different spellings/variations of potential covid illness visits/subject contacts.72 There are also 91 DESGTOR entries with unique 15-digit numerical codes of unknown significance, these are all prefixed POT_COVID_CONVA, POT_COVID_ILL, UNPL, or V202_SURVEIL_SWAB. Matching subject records between SV (subject visits, the most complete repository of covid illness visits as shown previously) and ADDV is likely to reveal potential unlogged adverse events, with the DESGTOR data point being an important additional qualifier.
A free-text visit description might not seem like much, but any clue is helpful when trying to better understand the protocol deviations. A number of them73 are inscrutable due to not being mentioned in the CRF or inadequate description.
Primadonna PIs
While there is barely a site without lengthy query/response volleys, and perhaps due to the language barrier or due to a refusal to be gaslit, the trial direction of one Turkish site failed especially; otherwise, there are very few and far between glimpses of Pfizer not getting their way, in form of a “RELATED” here or a refusal to delete an AE there. Not without substantial coaxing, however.
Subject 12091014 is a tremendous mess, with a positive covid illness from 19.-20.12., yet discharged from hospital on the 25.12., and the swab dated 31.12. The adverse event log has an event of covid-19 from 18.-23.12. The CRF also shows receipt of an influenza vaccine on 16.12. and Remdesivir while in hospital, along with obvious liver damage in the covid illness lab result forms.
Subject 12101026 is remarkable due to the site directly ignoring Pfizer’s clinical direction with regards to covid illnesses and adverse event reporting. 15.12., an entry by the site: “Patient had no covid-19 positive PCR test. So AE need to be recorded”, to which Pfizer replies the next day “Medical monitor: This is no correct. ACUTE UPPER RESPIRATORY TRACT INFECTIONS should be documented ONLY on the ILLNESS DETAILS CRF form irrespective of any SARS-CoV-2 test result and should not be captured on the AE CRF page” but the site remains steadfast, replying five days later with: “According to Safety team, the SAE onset date should be the date when the first signs/symptoms started, even if they were non-serious. The symptoms started on 07 Nov 2020 as non serious and the AE should be remain.” at which point Pfizer concedes, resulting in the subject having two adverse events of “acute upper respiratory tract infections” from 7.-16.11. and 16.11.-7.12.
Subject 12141039 contains a fantastic exchange between Pfizer and the site in the audit trail of an adverse event of diarrhea: upon multiple Pfizer queries demanding an illness visit be completed, the site replies with : “PI confirmed that this subject had only diarrhea due to eating foods that upset the digestive system and evaluated diarrhea as not COVID symptoms. This patient is not COVID-19.Not every symptom is COVID-19.” which garners the Pfizer response of “Medical monitor: During this study every symptoms listed Per Prot Sect 8.1. should have please triggered a potential COVID Illness Visit irrespective of perceived etiology or clinical significance. A protocol deviation will be documented”, which sets off another volley of queries and responses.
German site 1194 displays a natural affinity to the “covid illness” loophole, with each of the three supplied CRF files having adverse events hidden that way.
Subject 1002 has 11 days of chills after dose 4 hidden in covid_a.
Subject 1033 had 2 days of symptoms in December, yet when Pfizer asks for a convalescent visit, the site simply stonewalls by repeating “No Covid diagnosis. Therefor no Convalescent visit was performed” until Pfizer stops querying. There is a convalescent visit for cov_a in the CRF, but they took the serum samples from visit 101 (dose 3). This subject’s second covid illness had the diagnosis of “Immunnisation reaction.” with fever, muscle pain and the “other” symptom of lymphadenopathy until Pfizer demanded that adverse events be entered into the record, after which the diagnosis was deleted; only lymphadenopathy made it onto the adverse event log. The site didn’t make a fuss about the second “illness” convalescent visit.
Subject 1058 has the adverse events of lymphadenopathy, headache, injection site pain and arthralgia after dose 4, yet the CRF reveals a concurrent covid illness with the additional symptoms of fever and chills. Remarkably, all three Site 1194 CRFs are placebo patients who developed lymphadenopathy after real vaccination.
Site 1007’s PI never really seems to get the hang of lymphadenopathy causality: the first case of lymphadenopathy, inflicted on subject 10071050, is deemed “clinically significant” and thus worthy of inclusion on the adverse event log 21 days after being initially recorded. I’d like to highlight this remarkable site entry from 17.7.: “per PI:The adenopathy is not clinically significant. She has no pain with it & there is no alteration in function of the arm. But it is there & she noticed & brought to my attention. So, to me, that is an adverse event that is not clinically significant.” The adverse event referred to was created the day of the Phase 1 scheduled physical assessment one week after dose 2 with the lymph node finding and marked related, which Pfizer queries: “Please can you confirm in the PI's judgment why this AE is felt to be related to IP?”. Site responds later that day: “per PI:I called related because it is in the same arm as the vaccine and the area where the shot was given has lymph node drainage into the axillae”, and it takes Pfizer three days to answer with “Response satisfies query”.
The next site 1007 lymphadenopathy is handled differently. 1097’s bilateral cervical lymphadenopathy from 23.8.-3.9. with dose 2 being on 20.8., was marked not related to study intervention, but to “Possible viral syndrome”, yet the subject’s only covid visit is two months later.
10071117’s lymphadenopathy after unblinding was marked “related” without a fuss, and so was 10071159’s if you gloss over the two days additional days it took the site to complete the causality assessment data point, compared to the other fields on the form.
The same cannot be said of site 1231; of 25 lymphadenopathies, only 5 were deemed related. While the “related” events all occur 1-2 days post-vaccination, so do several “not related” ones; subjects 2080, 3674 and 4562 have lymphadenopathies starting 2-4 weeks after dose 2. One placebo subject has 3 days of lymphadenopathy after dose 2, another has two events from 23.12.-23.2., and the remaining three placebo cases are after unblinding.
There is plentiful evidence of inconsistent application of the protocol regardless of amendment; as described in the corresponding section, most “changes” were in fact re-wordings in order to better emphasize how the sponsor wanted the study conducted. What does not become clear is why some of the data points contested between site and sponsor differ between submitted CRF value and database value, and others don’t.
Assorted odds and ends
The missing letters bug and potential exploit A small number of CRF files74 are affected by what seems to be some form of export bug, resulting in single missing letters interspersed throughout the document, including in “pre-filled” fields. This is mostly innocuous, except for subject 10441152, where it leads to omission of the date of entry of the dose 2 visit, which is problematic as the reactogenicity eDiary assessment form, part of the visit as per schedule, is entered on 3.11., as compared to the vital signs entered on 19.10.
Subject 10381050’s CRF includes a Pfizer tip on how to deal with the unwieldy InForm eCRF program: “As reported before, DIRECT entry of the resolution date will change the information on the initial report; therefore, this change cannot be reported outside of comment section due to deficiency of CRF design. Please advise if any further actions needed.”
Subject 10281033’s CRF includes an offhand entry revealing that Pfizer’s safety database was offline for what seems like all of January and essentially all of February, too. After Pfizer complains that the last SAE report update was received on 21.12., the site replies on 16.2. with: “I have a fax confirmation that the EDP was sent on 02Feb2021 and the SAE follow up was sent in on 01Feb2021“, to which Pfizer replies a whole nine days later on the 25.2.: “Safety unit has confirmed updates are on hold as they resolve technical reporting problems“. How long was the database effectively offline? How did they work off the backlog while being inundated with post-marketing adverse events to the point of adding more than 1.500 safety personnel by June 2021?75
Subject 10841480’s CRF informs the reader in a site entry on 4.11. that the principal investigator is OOTO (presumably “out of the office”) until 11.11. This subject is also notable for having a heart attack 12 days after dose 1, yet receiving dose 2 on schedule seven days later, and being listed as BNT trial arm assignment in the CRF and placebo in the database.
Keeping with the topic of PI oversight from the previous subject, it seems as if site 1161 was not in fact excluded from the study, or at least that’s not the full story; all 43 subjects were excluded from safety analysis due to “Unreliable data due to lack of PI oversight”, yet both CRFs provided from that site state that the study was only paused. On subject 11611016’s dose 2 visit audit, Pfizer queries the 87-day delay: “PDQ: DOV at this visit is out of window (87 day) from DOV (01/Aug/2020) at V1D1VX1L. Please either update DOV or confirm whether visit is out of window due to temporary delay of vaccination/ else provide details as appropriate.“ to which the site replies: “visit 2 is oow due to study on pause per sponsor“. Similarly, subject 11611020’s CRF also alludes to “subject has not received vaccine for v2. study on hold” in response to a Pfizer query concerning the missing dose 2.
The adolescent CRFs
There are 18 CRFs included in the 12-15 production. With 2.307 adolescents enrolled throughout the course of the study, that equates to 0,76% of subjects getting a CRF, almost exactly one third of the CRF/subject rate amongst the 16+ population, which is a rather strong hint as to why there was such a strong focus on enrolling 16- and 17-year olds post-November data lock. There are 11 SAEs with Pfizer adverse event numbers in the 16+ database associated with adolescent subjects - only the placebo subjects have CRFs. Notably affected by this unexplained omission is Maddie de Garay’s CRF, subject ID 10071620.76
The adolescent production has its own database files, due to the data cut-off being 11.11.2021, as compared to the 16+ 1.4.2021. As mentioned previously, I’ll be looking at these subjects separately, but I felt this was too important not to mention.
Conclusion
The CRF review has revealed the testing of safety and efficacy intended by the clinical trial to be inherently flawed, if not sabotaged. The separate reporting of adverse events, solicited adverse events (reactogenicity) and potential covid illnesses fundamentally distorts the account of any given subjects’ health outcomes thorughout the trial, at best to the point of inaccuracy. There are hundreds of instances showing this distortion occurring at Pfizer’s direction; the frequency with which trial sites had to be prompted to separate adverse events and “symptoms of disease” demonstrates just how counter-intuitive the protocol was. There are numerous instances where the reactogenicity and/or illness symptoms “pools” were used to keep negative health outcomes off of the adverse event log.
The most important metric of the study, efficacy, is the most manipulated aspect of trial participation by a wide margin and marred by several sources of systematic error. The vaccine arm had more symptoms, yet fewer illness visits; when the seven-day embargo on covid tests post-dose was broken, it was overwhelmingly abused to keep health outcomes off of the adverse event log. There is also a clear and persistent bias against placebo in frequency of untested symptoms.
The significant proportion of potential illnesses created retroactively or only at sponsor request, along with less than 1/4 of subjects having a potential covid illness, reveals that there was not enough testing being done, especially in the first quarter of the trial. This is probably another undisclosed-as-problematic separate PI instruction, as is likely the case with gynecological medical history assessment; it stands to reason PIs were instructed to select potential cases to be tested carefully in the beginning of trial (perhaps due to the sparse capacities of the central lab, being clogged with tens of thousands of enrolment swabs?). Later on, Pfizer “flipped the script” and began aggressively querying adverse events for deletion in favor of potential covid illnesses, in order to reduce adverse events and artificially increase the testing rate.
It is noticeable that unfavorable causality assessments were only queried once, if at all, yet Pfizer proved to be remarkably obstinate in their clinical direction of adverse events, failing only a few times to get the site to do what they want. This direct “micromanaging” dispels any notions there might be of trial sites being truly independent. Pfizer schematically influenced subject management in real time.
Many of the trial sites were enrolling subjects for multiple covid vaccine sponsors at once. There is an especially large overlap with Moderna. This is an area of further study, I continually update my progress in this Google doc.
who writes the excellent Vaccine Data Science substack programatically compared the investigator files for Pfizer77 and Moderna78 and found a total of 22 sites involved in both trials.79 Notable inclusion is University of Texas Medical Branch SIVS Clinical Trials Program under Richard Rupp - Pei-Yong Shi’s lab at the same university was the main lab for several Pfizer-BioNTech C459 trials. There is a fascinating assortment of local news station segments about trial sites with cameos of their principal investigators. Many of the sites regularly change their names, or have changed their names since the covid trials. Many websites related to the covid trials are only on archive sites, and include details such as “up to 2000$ for participation in the Moderna study” or “60-120$ per site visit, depending on the study”. Many of these trial sites have since been involved in further studies by the same sponsors, which is unlikely to occur should there have been complications. The threat of recording a deviation often induced the desired site response - were there financial incentives for avoiding deviations, or penalties for incurring them? There are substantial differences in sponsor query behaviour between sites, as well as implausible variations in frequency of CRF-required subjects. Less than one third of potential covid illnesses having a diagnosis recorded is indicative of the selectively enforced and excessively complex data collection rules. There are frequent direct references to data cut-off deadlines, and reporting was demonstrably delayed to exceed these timepoints for crucially important events.Diarrhea is often involved in the most blatant fraudulent manipulations. Remarkably, Pfizer-BioNTech were the only covid vaccine trial sponsors to include diarrhea as a solicited systemic adverse event80, proving they were anticipating high incidences. Why were they the only sponsor to anticipate high incidence rates of diarrhea? That being said, diarrhea was a solicited adverse event for the GSK/Moderna combination trial 217670 which investigated Shingrix and GSK’s influenza vaccine along with Moderna’s half-dose booster.81 Further investigation is necessary.
The over-representation of covid illnesses and adverse events amongst the low amount of provided CRF files has concerning implications for the dataset as a whole; there are not enough events and they are blatantly inorganic in their distribution. The illness eDiary plays a remarkably large role in the sponsor’s control of study sites, demonstrating how unreliable data gathering actually was, if Pfizer had to rely the third-party eDiary records to learn of a subjects’ symptom report. As revealed by Stephanie de Garay, Maddie de Garay’s mother, the TrialMax system and site communication procedures left a lot to be desired82 - no free text field means the site personnel were absolute arbiters of data entered. With more than 200 avoided illness visits, there was no shortage of such decisions being made.
A systematic review of adverse events with matching of subject medical history and vital signs measurements could provide additional insight. The high prevalence of “-lithiasis”-related disorders along with pancreas and gallbladder findings is an adverse event profile I was unaware of and which has not nearly been communicated as extensively as items like myocarditis, lymphadenopathy or appendicitis. This also applies to GI-related events, albeit to a lesser degree; items like small bowel strangulation, hernias or diverticulosis are prevalent.
The most problematic findings are the discrepancies in trial arm assignment between CRF and database, and the large amount of adverse events never entered into the corresponding CRFs, yet present in the database. Whether a subject received vaccine or placebo is the most important data point to assess results by, and any error fundamentally calls into question the entire trial methodology. Especially troubling is that most of the discrepancies are corrected, implying there is a double-check system in place, which in turn puts the uncorrected errors in an even worse light. Similarly, the reluctance of many sites to enter health outcomes into the CRF in favor of reporting exclusively to Pfizer’s safety database effectively creates a situation in which SAEs are reported exclusively at Pfizer’s discretion. How many safety database submissions without CRF entries were made in total by sites, and how many of those submissions were then transferred to the database by Pfizer? How does it happen that some events are “forced” into the CRF by Pfizer queries, while others aren’t?
Personally, I learnt that taking physical notes was a bad idea and a mistake I won’t be repeating for Moderna, which is up next after a closer look at the Pfizer-BioNTech adolescent data. Another discovery I made is that notes aren’t all that useful to abbreviate or summarize if you rack up enough of them. What I have to improve for the future is use of software to support the reading. Manually looking up subjects in databases was a massive bottleneck and was not done anywhere near consistently enough, so there are a lot of fraudulent elements potentially missed, which became obvious when writing up narratives. While there is already plenty of problematic material, further work is required to ensure comprehensive coverage.
What are fruitful angles of attack? Beyond going over the existing material more systematically and consistently, there is for example the issue of swabs and barcodes. Most of the local swabs are entered into the database as “UNK”, yet have a “NEG/POS” value entered in the CRF. Identifying which local swab results were or were not entered into the database will provide additional epidemological and efficacy data, and give insight to internal Pfizer decision processes. The barcodes associated with samples are another vulnerability - especially the subjects who had barcodes entered in the CRF which dont appear in the database. Systematically evaluating when a data entry was made vs when the data was generated is also guaranteed to reveal anomalies, for example there are vaccination visits entered up to six days after the fact that I’m aware of. The issue of trial sites, their PIs and potential conflicts of interest needs a lot more scrutiny; due to the large overlap of Pfizer-BioNTech and Moderna trial sites, I expect the Moderna CRFs to reveal some interesting new info.
My goal was to convey not only the insights gained from reading the case reports, but also the impression I was left with. It could be shorter, but any summarization will lose fidelity, and the devil here is truly in the details - the calloused, clinical way subjects were used and discarded.
Acknowledgements
Pierre, for your unwavering dedication and relentless effort
Flo & Julia, for your infinite patience for my hermit-like behaviour these past months, and for keeping me sane(-ish)
HaJo Kremer, for starting me on this investigation, and for invaluable advice
Aaron Siri and ICAN/PHMPT/The Highwire for making this material available and being at the forefront of the good fight
Overview of the Excel file
Right-clicking these “< >” arrows at the beginning of the list will open a scrollable window with all sheets.
Excel instructions saying “search” or “filter” require inputting the search term into the text field that appears when you click the little triangle in the title row of a column.
Database file sheets
All worksheets between “crflist” and “IDList” are database files.
ADSL Analysis Dataset Subject Level, CO Comments, ADDS Analysis Dataset Disposition, CM Concomitant medication, MH Medical History, SV Subject Visits, DV Deviations, ADDV Analysis Dataset Deviations, IS Immunogenicity, HO Healthcare Occurrence/Utilization, ADAE Analysis Dataset Adverse Events, CE Clinical events
Non-database sheets
devlist - table necessary for the “Deviations” section listing CRF/non-CRF deviations
jpg + 1001-1007 - listing of JPG pages in CRFs that are not entirely JPG, with a separate sheet for sites 1001-1007
crf_site + crf_site 2 - site listings with additional metrics + beginning of Sheet 5 from the Google doc (only this link is up to date) with search results for PI/site names
subj_row_file - a file from OpenVAET listing databases, their descriptions, how many rows they have and how many unique subjects they concern
cov_a - a table for exploring the covid_a illness data entry permutations between SV/ADSYMPT/CE/MB
Tab1 - “junk” tab for list creation/filename separation
gfx - sheet for covid visit graphs
PA - overview of protocol amendments 0-15
IDList
I make extensive use of “COUNTIF”83 to pull datasets out of databases. Each column in the tab “IDList” has a predefined list of subjects which you can use to filter the databases.
List use example
Goal: All adverse events experienced by 16+ CRF subjects with a medical history item related to excessive female bleeding.
This requires two lists: a) all 16+ CRF subjects and b) subjects with a corresponding medical history item, listed in IDList columns T and AE. In the Excel file, open the tab “ADAE” and select any cell of the column “IDList”; it will contain the following formula:
=WENN(ZÄHLENWENN(IDList!EK:EK; D2)>0; "Ja"; "Nein")
In this case, it’s referring to the IDList column EK, which lists 85+ subjects. D2 after the semicolon inside the brackets in the above formula refers to the column in ADAE that contains the subject ID, this can vary between database files. Replace EK in the formula with T and hit enter.
Repeat the above step with the “IDList 2” column, by replacing whatever column is currently referred to with AE.
The IDList columns should now read
=WENN(ZÄHLENWENN(IDList!T:T; B2)>0; "Ja"; "Nein")
=WENN(ZÄHLENWENN(IDList!AE:AE; B2)>0; "Ja"; "Nein")
Filtering column IDList 1 to “Ja” will show only CRF subject adverse events, and filtering column IDList 2 to “Ja” will show only subjects with gynecological MH, so setting both to “Ja” will only show adverse events recorded for CRF subjects with bleeding related medical history.
One-stop adress on how to exploit and access the full spectrum of available database files:
crflist
One row per CRF subject. ADO - flag for 12-15 year-olds; cov_visit - had a potential covid illness y/n; swab - has a covid illness-associated swab; DV - has a deviation; vis_DV - has a covid illness visit-associated deviation, Reacto - S for severe, C for clean (no recorded events), Y for at least one recorded reactogenicity event, N for not part of subset, MH - number of database medical history items, EoSDT - end of study date, EoS - end of study reason, EoTDT - end of treatment date, EoT - end of treatment reason, vx1 vx2 vx3 vx4 - dose dates, dose1 dose2 dose3 dose4 - ACTARMCD value (actual trial arm), IT - has mention of which IT system, Reminders - are certain standardized “reminder” phrases present in the CRF, Terms - are certain terms present, flu - did the subject receive a flu jab, cov_vis ND - not done covid illness visits and denied subject eDiary symptom reports, deleted Aes - deletions in the adverse event report form, AE not in CRF - adverse event present in the database yet not in the CRF, MH edits - when were medical history items edited/added, cov_vis Abuse - evidence of abuse of potential covid illness/adverse event reporting dilemma, placebo? - subject’s placebo assignment is questionable SAE flag - adverse event descriptor/CRF inclusion flag EDP - exposure during pregnancy, reacto error - reactogenicity subset assignment error, v/p error - trial arm assignment error, narrative - short summary of CRF, note - keyword or analysis specific detail, example - classification
“the spicy bit: there is a database file called "CO" for comments, which includes rationales for not done tests. hundreds of "per PI" unswabbed possible cases, then pfizer changes some to covid visits, but how are you gonna swab months later?” -https://x.com/a_nineties/status/1773146525096456556
Excel: Sheet “CO”, column “TAG”=“NDSPONS” “NDSUB” “NDU” “NDERR” “NDTIME” “NEG” “NDTELE” “NDSUBREP” “NDSITE” “ND”
Crf_master, 458 mb - https://mega.nz/file/bZYE0BpB#3aVCs3hBORpIZRG8KWZ-sHyiM9xpyEboGF4cd0XIuJg
See section this section for further details https://modarnlife.substack.com/i/146474910/overview-of-the-excel-file
Local solicited AEs: Pain at the injection site, Redness at the injection site, Swelling at the injection site
Systemic solicited AEs: Chills, diarrhea, fatigue, fever, muscle pain, joint pain, headache, vomiting
PHMPT.org - Module 1.6.3 Correspondence Regarding Meetings https://phmpt.org/wp-content/uploads/2023/10/125742_S1_M1_meeting-correspondence.pdf
Page 476 meeting-correspondence.pdf
Page 574 meeting-correspondence.pdf


Subjects with lymph-related AEs with localisation and no CRF: 10161057 10831197 11171071 11281344 11281344 11301051 11411179 11691046 11701137 11701450 11421003 11791096 12141051 12231245 12261164 12611064 12641181 12651082 10901078
Excel: Database ADAE, AGE≠12,13,14,15, column “IDList 1”=Y (default setting is CRF subject yes/no, IDList column T), search column “AEDECOD” for “lymph”, deselect cancers/lymphopenia/-cytosis
Subject 10061161 (29/F/Placebo) has an AE of “Maternal Exposure before Pregnancy” on 28.12. There is no corresponding CRF file.
Database ADAE, AEREFID=not empty, AGE≠12,13,14,15
Excel: Database SV, AGE≠12,13,14,15, VISIT=COVID_A-F, CRF=Y/N 478 of 10.476 covid visits, VISIT=COVID_A1-F1, CRF=Y/N, 390 of 8.693 convalescent visits.
Database ADAE, AGE≠12,13,14,15, CRF=Y/N 2766 of 37078 adverse events.
ICANdecide.org, “According to FDA, this is the final batch of documents related to the authorization of the Pfizer vaccine for ages 12-15.” https://icandecide.org/press-release/new-case-reports-released-for-pfizer-ages-12-15-and-moderna-ages-18-show-myocarditis-appendicitis-intestinal-perforation-and-more/
This previously said 98, hat-tip to HaJo Kremer for catching this error!
CRF Subjects discontinued before dose 1 (5): 11221026 12051028 12311409 12311926 12481218
Excel: Sheet “crflist”, search column “vax1” for “NA”
This includes efficacy endpoint-conforming (positive central lab swab) illnesses and adverse events involving covid.
Cardiac events including hypertension, Afib, angina etc.
Kidney, gallbladder and other “stones” cholelithiasis, cholecystitis, nephrolithiasis, pyelopnephritis etc.
Strokes, TIAs, aneurysms, haemmorhages (including gastric), clots, embolisms, stenoses
Upper respiratory infections (URI), urinary tract infections (UTI), pneumonia, sepsis, abscesses, COPD, asthma
Appendicitis, gastroenteritis and GI-related AEs, colitis, diverticulitis, intestinal hernias, bowel obstructions etc.
All “osteo-” AEs like osteoarthritis, osteopenia etc. including vertebral hernias
Sites which supplied no CRF files to the primary series submission: 1002 1024 1073 1179 1202 1208 1219 1258 1269
Excel: Sheet “crf_site”, search column “# of CRFs” for “0”
Sites with a %CRF/%subj ratio < 0.6: 1179 1269 1024 1073 1002 1258 1219 1208 1202 1056 1098 1123 1229 1177 1224 1052 1149 1126 1162 1163 1036 1066 1008 1139 1170 1121 1168 1101 1011 1147 1230 1133 1009 1171 1197 1094 1265 1057 1037 1157 1088 1046 1232 1084 1248 1235 1270 1135 1261 1030
Excel: Sheet “crf_site”, sort column “%CRF/%subj” in ascending order
Sites with a %CRF/%subj ratio > 1.4: 1226 1114 1110 1223 1210 1231 1015 1068 1019 1247 1140 1246 1128 1214 1205 1251 1003 1131 1169 1095 1055 1118 1220 1161 1178 1209 1217 1252 1117 1122 1145 1111 1129 1260 1195 1081 1097 1218
Excel: Sheet “crf_site”, sort column “%CRF/%subj” in descending order
PHMPT.org - C4591001 Month 6 Protocol - Page 143 - discussed in greater detail in “the protocol amendments” section
Overlapping solicited systemic adverse events and potential covid illness symptoms: headache, fatigue, fever, chills, diarrhea, vomiting
PHMPT.org - CRFS-for-site-1081 Page 408
Subject CRFs with clear signs of fraud related to potential covid illnesses and visits (140):
10011093 10031113 10031186 10051054 10051293 10051387 10061040 10061052 10071097 10071306 10091128 10121163 10131176 10131190 10131699 10131718 10131786 10151238 10161199 10161289 10191145 10271105 10371052 10381050 10381051 10391038 10421023 10421217 10441194 10481032 10541173 10571052 10681079 10771226 10801006 10811026 10811036 10811045 10811110 10811135 10811194 10831060 10831162 10841538 10851018 10871150 10871228 10871286 10881139 10901300 10901415 10911197 10921187 10921208 10941112 10951101 10951228 10961031 10961181 10971011 10971017 10971061 11091448 11101236 11111010 11111103 11161045 11161224 11171036 11171086 11281103 11281296 11291127 11291183 11301081 11311151 11311204 11361082 11401009 11401117 11411153 11411221 11421044 11471230 11521095 11611020 11661047 11691056 11781061 11941033 11941058 11951017 11951023 11951132 12181001 12231058 12231159 12261094 12261210 12261338 12261599 12262052 12262089 12262253 12262272 12311118 12311295 12311352 12311766 12311862 12312378 12312390 12312982 12313496 12313653 12313674 12313785 12313879 12313998 12314001 12314128 12314759 12315520 12315622 12315632 12411051 12411067 12411269 12461070 12471066 12471121 12481120 12511029 12511060 12521011 12601018 12651101 44441422 44441473 44441550
Excel: Sheet “crflist”, column “cov_vis abuse”, hide empty cells
BNT subjects with missed/not done covid visits (92): 10011093 10031065 10051214 10051411 10061094 10081337 10121163 10131190 10131386 10151089 10151101 10151238 10191002 10191010 10371052 10471114 10561022 10571065 10681079 10771194 10771226 10801035 10841266 10841317 10851018 10871286 10881139 10951101 10951204 10961017 10971064 11091111 11101160 11101164 11101236 11161059 11161131 11171058 11171086 11201266 11201350 11221053 11271023 11281123 11281359 11291046 11291127 11311021 11401002 11401244 11411143 11461109 11461152 11521411 11521450 11671175 11781073 12041023 12261043 12261282 12261769 12262089 12301025 12311118 12311295 12311766 12312390 12312885 12313496 12313674 12314035 12314216 12314921 12315291 12315520 12315579 12321159 12411343 12411915 12411978 12412035 12461070 12511029 12511033 12511060 12511239 12511262 12521010 12601037 44441161 44441249 44441550
Excel: Sheet “crflist”, set column “cov_vis ND” to hide empty cells, set column “vx1” to “BNT162b2”
Placebo subjects with missed/not done covid visits (61): 10081056 10941155 10071280 12314759 11461191 10801006 12231252 12261241 10961036 11951059 11951131 12312893 11171141 11091036 12481120 10961056 11251154 12315622 10191226 12201044 12262253 11091074 11781300 11951017 12411197 10371214 12315301 10131255 10951197 11521095 12141039 11521222 11161045 12411887 12031026 11701090 10441152 12411829 12314372 10031111 12311901 10911197 11101187 12411679 10301059 11471007 12312722 12314197 11291183 11501084 11301031 12651101 10061040 10801013 10191146 12315324 10281059 10061052 11181044 12311834 11291037
Excel: Sheet “crflist”, set column “cov_vis ND” to hide empty cells, set column “vx1” to “Placebo”
Placebo subjects with first missed/not done covid visit after unblinding: 10081056 10941155 10071280 11461191 10801006 12261241 11951059 11951131 11171141 12481120 10961056 12201044 11781300
Excel: Sheet “crflist”, search column “cov_vis ND” for “after vx3”
Deviations related to missed covid visits/swabs:
Lab performed out of window; Procedure/Test not done; Visit not done; Nasal swab not collected for the visit where it is required; Visit performed outside of protocol specified window; Potential COVID-19 illness visit not done when required per protocol; Procedure/Test not performed per protocol
487/1028 CRF files have no adverse event reminders. The term “safety database but missing in AE CRF” included in the default query formulation appears in 461/1028 CRFs. The term “not reported to safety database” appears in 499/1028 CRFs.
This was initially reported as part of point 68 in https://openvaet.substack.com/i/144275433/further-evidence-of-data-manipulations-on-safety-data-recordings
Excel: sheet “crflist”, column “v/p error”, hide empty cells
Excel: sheet “crflist”, column “placebo?”, hide empty cells
Placebo CRF subjects with unblinding dates but no dose 3: 10281059 10061020 10371318 10461175 10471194 10541173 10821149 10831050 10841480 10951197 11091448 11141075 11201002 11201432 11211112 11471230 11561044 11621059 12231252 12312893 12313437 12461110 12471066 12511072
Subjects erroneously included in narratives and thus CRF submission: 10371141 10901492 10911297 10921021 12541006 12541189
C4591001 Six-Month Narratives - 125742_S1_M5_5351_c4591001-interim-mth6-narrative-sensitive.pdf Page 713: “After the data cutoff date of 13 Mar 2021 (with database snapshot taken on 25 Mar 2021), it was determined that Subject C4591001 1037 10371141 was not pregnant and remains in the study to be evaluated for safety, immunogenicity, and efficacy.”
Page 291: “After the data cutoff date of 13 Mar 2021 (with snapshot taken on 25 Mar 2021), it was determined that Subject C4591001 1090 10901492 did not withdraw from the study because of the fatigue and remains in the study.”
Page 301: “After the data cutoff date of 13 Mar 2021 (with snapshot taken on 25 Mar 2021), it was determined that Subject C4591001 1091 10911297 did not withdraw from the study because of the fatigue and remains in the study.”
Page 305: “After the data cutoff date of 13 Mar 2021 (with snapshot taken on 25 Mar 2021), it was determined that Subject C4591001 1092 10921021 did not withdraw from the study because of the pain and remains in the study.“
Page 485: “After the data cutoff date of 13 Mar 2021 (with database snapshot taken on 25 Mar 2021), it was determined that Subject C4591001 1254 12541006 did not withdraw from the study because of the chills and remains in the study.“
Page 494: “After the data cutoff date of 13 Mar 2021 (with database snapshot taken on 25 Mar 2021), it was determined that Subject C4591001 1254 12541189 did not withdraw from the study because of the headache and remains in the study.“
Excel: Sheet “crflist”, search column “cov_vis ND” for “after vx3”
Excel: ADDS database, column “DSDECOD”= ”PHYSICIAN DECISION”, AGE≠12,13,14,15, CRF=Y/N
Full JPG CRFs: 11101236 10971011 10971064 10971023 12411347 44441634 10911002 10911170 10911213 11231381 11351257
Excel: Sheet “crflist”, search column “example” for “JPG CRF”
Excel: Sheet “crflist”, search column “example” for “JPG”. Also see sheet “1001-1007” for a breakdown of the corresponding JPG pages.
To be added once published
“Please submit fully functional pdf documents (e.g., that can be searched to locate information) for the following reports that are hyperlinked from the BLA submission (document bnt162-01-interim3-report body):
a. R&D Report R-20-0253 (28 November 2020)
b. R&D Report R-20-0235 (27 Nov 2020)
c. Interim report GA-RB-02201A (19 March 2021)
d. R-20-0244 (19 March 2021)
e. Interim Clinical Study Report R-20-0241 (20 March 2021)”
Submission 1 file with JPG data (73 mb) - released to PHMPT October 2023 - https://pdata0916.s3.us-east-2.amazonaws.com/pdocs/100123/125742_S1_M5_5351_bnt162-01-interim3-reports.pdf
Submission 7 file with machine-readable data (25 mb) - released to PHMPT April 2023 - https://phmpt.org/wp-content/uploads/2023/04/125742_S7_M5_5351_bnt162-01-interim3-reports.pdf
PHMPT.org - 125742_S1_M5_c4591001-S-csdrg.pdf Annotated CRF section starts p17/86
PHMPT.org - Annotated Study Book - C4591001
Subjects with mention of IT system:
Florence (37): 10121112 10191071 10371052 10371141 10371214 10371318 10421023 10481032 10551176 10681091 10831029 10871029 10901300 10901384 10971017 10971025 10971084 11101164 11111010 11111103 11111109 11301031 11341378 11401306 11561006 11561160 11611020 11781061 11781138 11781293 12181001 12181012 12211002 12511050 12601018 12601075 12601108
Redcap (4): 10301059 12312914 12315429 44442304
Argus (12): 10031113 10131084 10131176 10811026 10851216 10951107 10961062 11141006 11281009 11451063 12231159 12291083
Impala (55): 10031038 10051411 10071117 10071280 10281003 10391038 10391075 10441093 10801152 10841470 10891289 10901300 10951101 10961056 10971061 10971087 10981024 11071044 11091084 11161045 11161253 11221026 11281192 11291005 11291037 11311095 11311222 11341174 11361082 11401035 11401306 11411153 11411221 11421202 11421247 11461302 11491313 11521450 11621059 11621128 11661078 11741042 12051028 12171031 12261707 12313496 12321213 12321293 12411468 12471244 12481218 12521011 12541014 12601128 12701069
Complion (9): 10811026 10811061 10811110 10811135 10951101 11171036 11281009 11281014 11281103
Trialmax (10): 10011093 10071280 11271112 11281267 11361029 11401117 12311295 12471226 12601018 12601037
Excel: search column “IT” for keywords
Physical binder quotes:
Subject 12181057, Page 117: “I need to delete all entries for this Vax 2 visit. I inadvertently entered the data from another participant's binder into this participant's Vax 2 fields.”
Subject 12601075, Page 173: “Sorry just confused, do you want the hospitalization details recorded here in inform somewhere or just have it in our physical binder?”
Excel: search column “note” for “physical”
14.3.2023, https://x.com/IamBrookJackson/status/1635727033748815872:
“Lawsuit Update: Judge Truncale awarded Pfizer with another 31 days of delayed discovery. PS. Dang,
! I thought I was jaded. Whoever you pissed off at
is vicious! She asked me to pass along 2 words & a pic: Argus Safety”
Subjects enrolled after 14.11. not from sites 1218, 1219, 1220, 1221: 11231424 11231435 11231436 11251245 11421320 11471260 11471276
Excel: sheet “MB”, column “VISIT”= V101_VAX3, V102_VAX4
Excel: sheet “SV”, column “VISIT”= V101_VAX3, V102_VAX4
Excel: sheet “IS”, column “VISIT”= COVID_A1, COVID_B1, column “ISMETHOD”= “N-binding Antibody Assay”
Anti-nucleocapsid antibodies following SARS-CoV-2 infection in the blinded phase of the mRNA-1273 Covid-19 vaccine efficacy clinical trial
Dean Follmann, Holly E. Janes, Olive D. Buhule, Honghong Zhou, Bethany Girard, Kristen Marks, Karen Kotloff, Michaël Desjardins, Lawrence Corey, Kathleen M. Neuzil, Jacqueline M. Miller, Hana M. El Sahly, Lindsey R. Baden
medRxiv 2022.04.18.22271936; doi: https://doi.org/10.1101/2022.04.18.22271936
Now published in Annals of Internal Medicine doi: 10.7326/m22-1300
[FDA:] “In the IS dataset, there is missing immunogenicity data for at least 12 subjects who received the 30 µg dose of BNT162b2. Please submit all immunogenicity data for participants who received 30 µg dose of BNT162b2 to the BLA.”
[Pfizer:] “These data were not available at the time of immunogenicity cut-off for this report because the samples were put on hold due to necessary testing prioritizations at the Pfizer labs (eg C4591001 6-month stability and booster; C4591007). Pfizer has now resumed testing of these samples and an updated BNT162-01 study report will be provided once it is available. Pfizer/BioNTech do not believe these data to be material to the review of the Biologics License Application.”
There is a sixth “potentially life-threatening” reactogenicity event in a subject under 16, so that CRF might be in the 12-15 production.
Excel: sheet “CE”, column “CECAT”=”REACTOGENICITY”, column “DUR”≠XX,0,1,2,3,4,5,6,7
Subjects with reactogenicity events with start date and no end date (CRF subjects in bold): 10161087 10811026 11201350 12171044 12461025 10011111 11201408 10051038 10051075 10071185 10191004 10441238 10441240 10441244 10791021 10791083 10791112 10841117 10841147 11201250 11471009 10901065 10901078 10961021 11101072 11181003 11181005 11181006 11201085 11201225 11201238 11201262 11201275 11201276 11201315 11201339 11201400 11201404 11201435 11231080 11231086 11271006 11271028 11311265 11391090 10871045 10901127 11281054 11421015 11491012 11611029 11621015 11701385 11851005 12031058 12311152 12311499
Excel: Sheet “ce”, column settings: 12-15=“No”, CECAT= REACTOGENICITY, CESTDTC=not empty, CEENDY=empty
DiffPDF, 20-day freeware license, powerful PDF comparison tool - https://www.qtrac.eu/diffpdf.html
PHMPT.org - Six-Month Interim Clinical Study Report C4591001 April 2021 - Each data table contains the information “(Cutoff Date: 13MAR2021, Snapshot Date: 25MAR2021)”
x.com - https://x.com/a_nineties/status/1717580660829757489 - “c4591001 was supposed to be phase1/2 only, they even designed "C4591002" as the corresponding phase 2/3, but CBER wanted them do keep it in one study. BNT162-01's placebo-controlled part b was always gonna get cancelled”
PHMPT.org - https://phmpt.org/wp-content/uploads/2023/10/125742_S1_M1_meeting-correspondence.pdf - Page 31, Type C Meeting Briefing Document dated 10.6.2020: ”Pfizer now proposes to replace Stage 3 of C4591001 in its entirety with the Phase 2/3 components previously outlined as C4591002, with changes based on CBER review.”
“Potential COVID-19 illnesses and their sequelae that are consistent with the clinical endpoint definition should not be recorded as AEs. These data will be captured as efficacy assessment data only on the relevant pages of the CRF, as these are expected endpoints.
Potential COVID-19 illnesses and their sequelae will not be reported according to the standard process for expedited reporting of SAEs, even though the event may meet the definition of an SAE. These events will be recorded on the COVID-19 illness pages in the participant’s CRF within 1 day.
Potential COVID-19 illness events and their sequelae will be reviewed by a group of internal blinded case reviewers. Any SAE that is determined by the internal case reviewers NOT to meet endpoint criteria is reported back to the investigator site of incidence. The investigator must report the SAE to Pfizer Safety within 24 hours of being made aware that the SAE did not meet endpoint criteria. The investigator’s SAE awareness date is the date on which the investigator site of incidence receives the SAE back from the internal case reviewers“
“Potential COVID-19 illnesses and their sequelae that are consistent with the clinical endpoint definition should not be recorded as AEs. These data will be captured as efficacy assessment data only on the relevant pages of the CRF, as these are expected endpoints.
Potential COVID-19 illnesses and their sequelae will not be reported according to the standard process for expedited reporting of SAEs, even though the event may meet the definition of an SAE. These events will be recorded on the COVID-19 illness pages in the participant’s CRF within 1 day.
NOTE: However, if either of the following conditions applies, then the event must be recorded and reported as an SAE (instead of a disease-related event):
The event is, in the investigator’s opinion, of greater intensity, frequency, or duration than expected for the individual participant.
OR
The investigator considers that there is a reasonable possibility that the event was related to study intervention.
Potential COVID-19 illness events and their sequelae will be reviewed by a group of internal blinded case reviewers. Any SAE that is determined by the internal case reviewers NOT to meet endpoint criteria is reported back to the investigator site of incidence. The investigator must report the SAE to Pfizer Safety within 24 hours of being made aware that the SAE did not meet endpoint criteria. The investigator’s SAE awareness date is the date on which the investigator site of incidence receives the SAE back from the internal case reviewers.“
Side-by-side comparison of PA 3 & 4:
The full Pfizer product transmission paragraph:
”Suspected transmission via a Pfizer product of an infectious agent, pathogenic or nonpathogenic, is considered serious. The event may be suspected from clinical symptoms or laboratory findings indicating an infection in a patient exposed to a Pfizer product. The terms “suspected transmission” and “transmission” are considered synonymous. These cases are considered unexpected and handled as serious expedited cases by pharmacovigilance personnel. Such cases are also considered for reporting as product defects, if appropriate.”
Excel: IDList “EK”
DESGTOR values referring to covid illness visits, values with a numerical coding are excluded: COVID_A, POT_COVID_ILL, COVID_B, COVID_C, POT_COV_ILL, Unschedule Teleheath Visit, Unplanned, Unscheduled Telehealth Visit, COVID-19 Illness Visit, COVID A, UNPL, COVID Illnes, AE, COVID_ILL, POT_COVID_IL, POT_COID_ILL, PO_COVID_ILL, COVID-A, POT_COVD_ILL, POT_COVID_ILL VISIT, COVID_D, COVID Illness Visit, POT_COVID_ILL_A, COVID_E, COVID Illness, Unplanned1, POT-COVID_ILL, POL_COVID_ILL, POT_COVID_ILL_B, UNPL_VAX, COVIDA, POT_ILL_VISIT
Deviations with unclear significance and/or not recorded in CRF documentation: IP administered prior to Almac authorizing it for use, IP Documentation Error - Confirmation of Treatment - Received Form errors, IP documentation error – IRT Confirmation Report error, IP verification incomplete, Other IP deviation, IP documentation error - Confirmation of blind error, Other lab deviation, Procedure not recorded in source documentation as having been performed, Randomization done out of sequence, Procedure not performed by a medically qualified individual at specified visits, Other visit deviation, Site staff training missing/incomplete
CRF files with random missing letters: 10441093 10441111 10441152 10801006
“number of additional employees for AE recording keeps going up, they were 100 shy of their 1800 june target by may and projected 300 more in the next 3 months” - https://x.com/a_nineties/status/1716147187137085469
PHMPT.org - Summary Monthly Safety Report March 2021 - Page 4822 - “Pfizer has also taken a multi-level approach to help alleviate the large increase of adverse events. This includes significant technology enhancements, and process and workflow solutions, as well as increasing the number of data entry and case processing colleagues. To date, Pfizer has onboarded approximately 600 additional full-time employees (FTEs). More are joining each month with an expected total of more than 1,800 additional resources by the end of June 2021.”
PHMPT.org - Summary Monthly Safety Report May 2021 - Page 7433 - “The root cause of late case completion is identified as the unprecedented volume of cases received consequent to the unprecedented subjects’ exposure to the vaccine. Pfizer commenced a program of measures to increase PV system capacity in preparation for launch including an increase in case processing resources. Pfizer has completed the onboarding and deployment of 1700 resources into the case management systems and anticipate this number to increase to approximately 2000 within Q3 2021.“
Del Bigtree’s The Highwire has an almost two-hour documentary on Maddie’s case:
https://thehighwire.com/ark-videos/rigged-maddies-story/
You can see her 9:15 US Senate testimony here: https://thehighwire.com/videos/stephanie-and-maddie-de-garay-testimony/
PHMPT.org - 16.1.4.1 LIST OF INVESTIGATORS AND NUMBER OF SITES AND SUBJECTS BY THE COUNTRY - https://phmpt.org/wp-content/uploads/2023/10/125742_S1_M5_5351_c4591001-interim-mth6-investigators.pdf
PHMPT.org - List of investigator sites for mRNA-1273-P301 - https://phmpt.org/wp-content/uploads/2024/05/125752_S3_M1_list-of-investigator-sites-p301.pdf
Principal investigators/sites listed for both Pfizer-BioNTech and Moderna clinical trials: 1006 1009 1012 1018 1042 1048 1082 1083 1084 1093 1095 1109 1114 1117 1118 1120 1123 1129 1133 1136 1142 1157
X.com - https://x.com/a_nineties/status/1712094802223722997 - Comparison of solicited adverse events between sponsors Pfizer-BioNTech, Moderna, AstraZeneca, Janssen, and Novavax.
x.com - https://x.com/a_nineties/status/1808227252359831850 - Thread with brief overview and link to final study reports for Sanofi/Moderna and GSK/Moderna combination trials QHD00028 and 217670
“Just so everyone is aware, there was NO free form available in TrialMax which is what was used in the Pfizer Trials. You had to CALL the Principal Investigator. Yes, we have emails and recorded calls but there is virtually no way to easily collect this data. There is a reason they did this.“ - https://x.com/shdegaray73/status/1758694646153183542
ZÄHLENWENN corresponds to COUNTIF. Please dont ask me why function names are localized in Excel, it’s an abomination.